A Levels Chemistry (9701)•9701/14/M/J/25
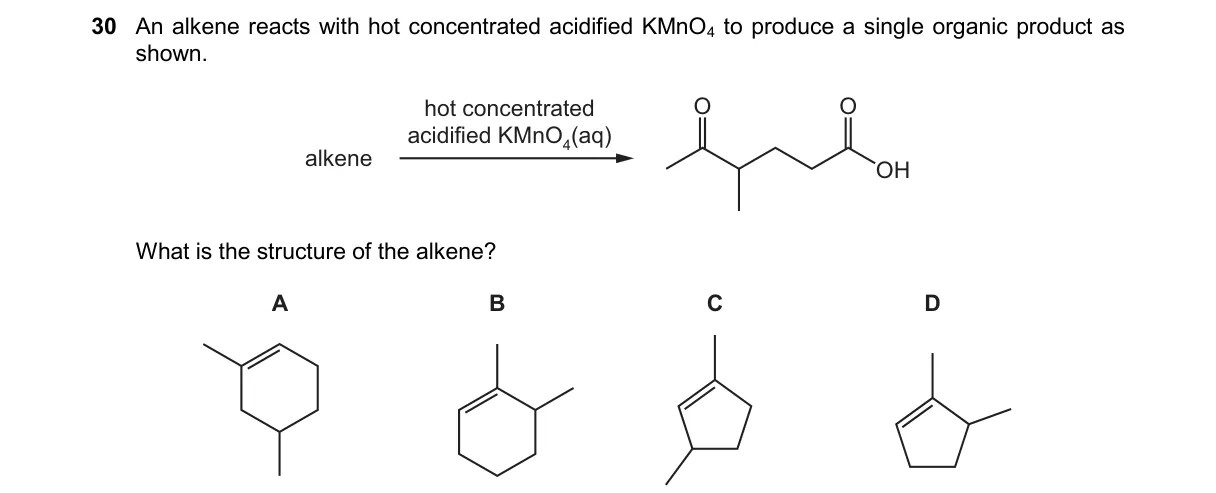
Explanation
Oxidative Cleavage of Alkenes with KMnO4
Steps:
- Hot concentrated acidified KMnO4 cleaves the C=C bond oxidatively.
- Each carbon of the double bond becomes a carbonyl: =CH2 to CO2, =CHR to RCOOH, =CR2 to RCOR.
- A single product indicates a symmetrical alkene where both sides yield identical fragments.
- Identify the alkene that, upon cleavage, gives one unique carboxylic acid or ketone.
Why D is correct:
- Option D shows a symmetrical alkene (e.g., 2-butene) that cleaves to two identical acetic acid molecules, matching the single product.
Why the others are wrong:
- A: Unsymmetrical alkene yields two different products (e.g., formic and acetic acid).
- B: Terminal alkene produces CO2 and a distinct carboxylic acid, not single organic product.
- C: Disubstituted unsymmetrical alkene gives a ketone and carboxylic acid mixture.
Final answer: D
Topic: Hydrocarbons
Practice more A Levels Chemistry (9701) questions on mMCQ.me