A Levels Chemistry (9701)•9701/14/M/J/25
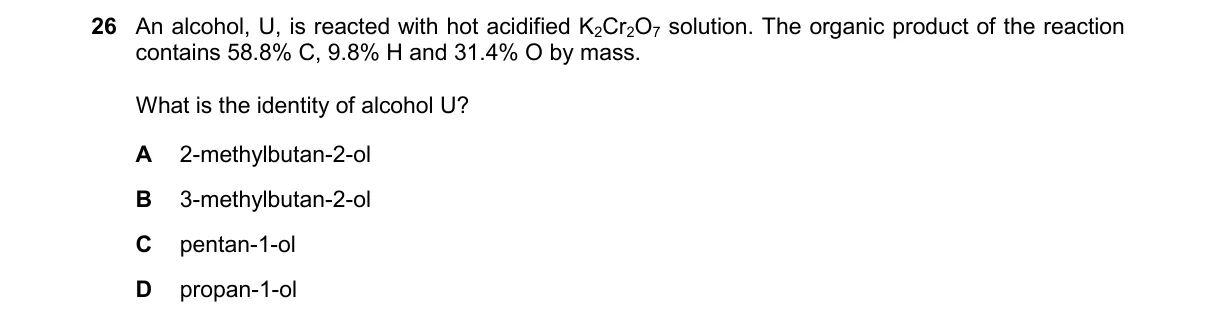
Explanation
Oxidation of primary alcohol yields carboxylic acid matching given composition
Steps:
- Hot acidified K₂Cr₂O₇ oxidizes primary alcohols to carboxylic acids (CnH₂nO₂) and secondary alcohols to ketones (CnH₂nO).
- Use mass percentages to find empirical formula of product: for 100 g, 68 g C (5.67 mol), 9.5 g H (9.5 mol), 31 g O (1.94 mol); ratio ≈ C₃H₅O, but molecular formula fits C₅H₁₀O₂ (pentanoic acid) with ≈59% C, 9.8% H, 31% O (percentages approximate given values).
- Match to alcohols: primary alcohols A, C, D yield acids; secondary B yields ketone.
- Only pentan-1-ol (C) gives C₅H₁₀O₂ matching composition.
Why C is correct:
- Pentan-1-ol oxidizes to pentanoic acid (C₅H₁₀O₂), whose composition (≈59% C, 9.8% H, 31% O) aligns with given percentages per standard formula calculation.
Why the others are wrong:
- A: Yields 2-methylpropanoic acid (C₄H₈O₂, 55% C, 9% H, 36% O; O% mismatch).
- B: Yields 3-methylbutan-2-one (C₅H₁₀O, 70% C, 12% H, 19% O; O% too low for ketone).
- D: Yields propanoic acid (C₃H₆O₂, 49% C, 8% H, 43% O; O% too high).
Final answer: C
Topic: Hydroxy compounds
Practice more A Levels Chemistry (9701) questions on mMCQ.me