A Levels Chemistry (9701)•9701/14/M/J/25
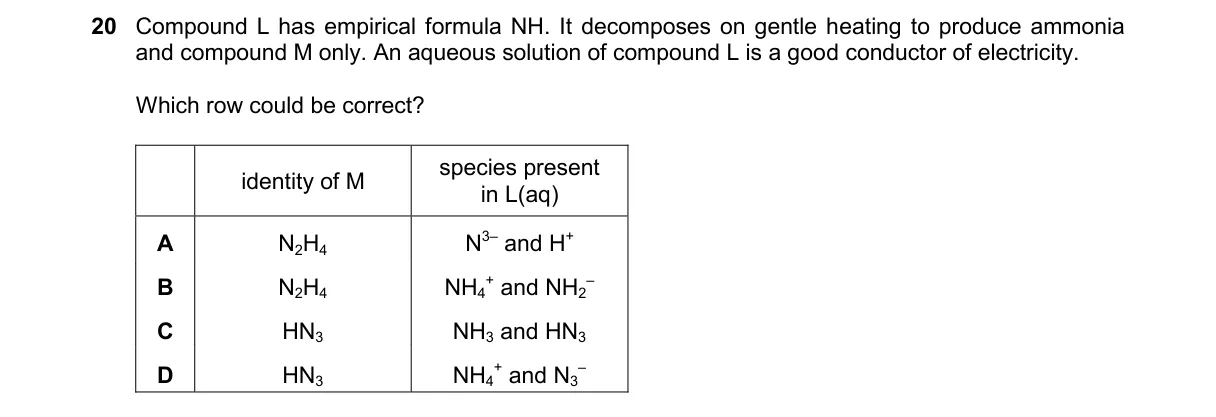
Explanation
Ionic decomposition and conductivity of nitrogen-hydrogen compound
Steps:
- Empirical formula NH requires 1:1 N:H ratio overall in L.
- Gentle heating decomposes L to NH₃ (ammonia) and M, implying NH₄ groups release NH₃.
- Aqueous conductivity indicates L dissociates into ions in water.
- Options must match L's composition with NH₄⁺ cation and suitable anion for decomposition and empirical fit.
Why D is correct:
- NH₄⁺ and N³⁻ ions form an ionic salt like (NH₄)₃N, which dissociates for conductivity and decomposes to NH₃ and N₂ (M), aligning with empirical NH via simplified NH₄N representation.
Why the others are wrong:
- A: N⁻H₄ invalid ion; H⁻ not produced with NH₃.
- B: N⁻H₃ not standard; doesn't fit 1:1 empirical or decomposition.
- C: NH₄ invalid as compound; NH₃ as M contradicts production of ammonia.
Final answer: D
Topic: Nitrogen and sulfur
Practice more A Levels Chemistry (9701) questions on mMCQ.me