A Levels Chemistry (9701)•9701/13/M/J/25
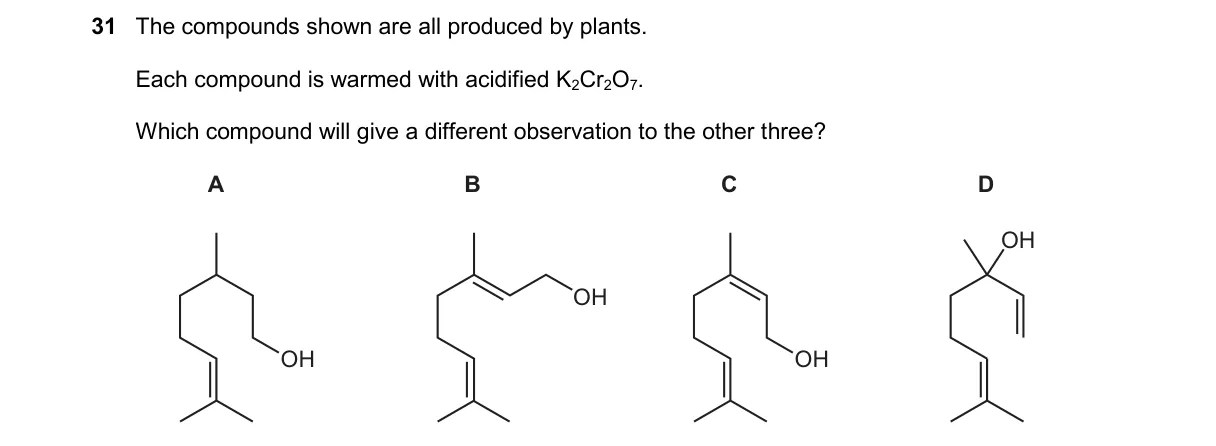
Explanation
Oxidation test with acidified K2Cr2O7 distinguishes alcohol types
Steps:
- Acidified K2Cr2O7 oxidizes primary/secondary alcohols and aldehydes, changing from orange to green.
- Plant compounds A, B, C are primary or secondary alcohols, undergoing oxidation with color change.
- Compound D is a tertiary alcohol, resisting oxidation.
- Thus, D shows no color change, differing from the others.
Why D is correct:
- Tertiary alcohols lack a hydrogen on the carbon bearing the OH group, preventing oxidation per the mechanism requiring H abstraction.
Why the others are wrong:
- A: Primary alcohol oxidizes to aldehyde then carboxylic acid, causing color change.
- B: Secondary alcohol oxidizes to ketone, causing color change.
- C: Aldehyde oxidizes to carboxylic acid, causing color change.
Final answer: D
Topic: Hydroxy compounds
Practice more A Levels Chemistry (9701) questions on mMCQ.me