A Levels Chemistry (9701)•9701/12/M/J/25
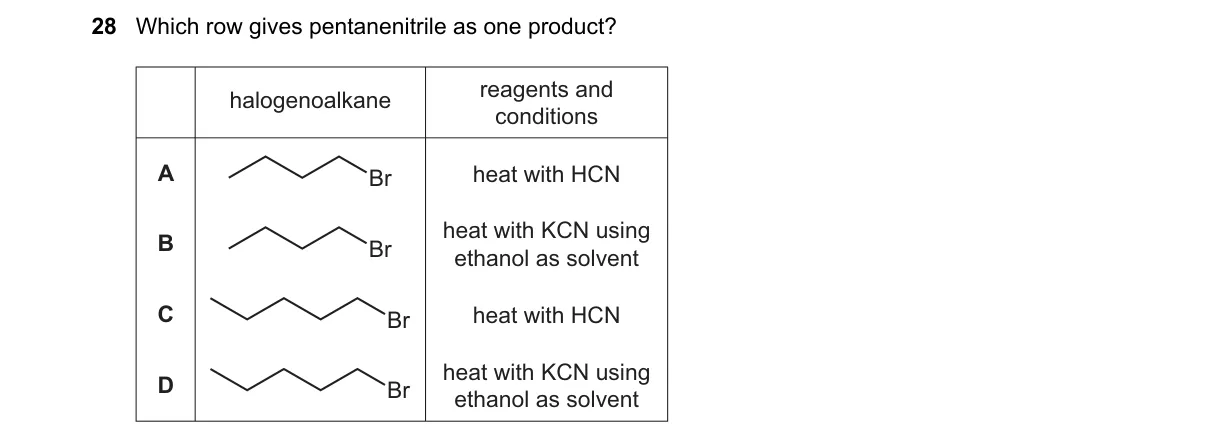
Explanation
Alcoholic KCN enables SN2 formation of nitriles from primary alkyl halides
Steps:
- Identify pentanenitrile (CH3(CH2)3CN) forms via SN2 reaction of 1-bromobutane (CH3(CH2)3Br) with CN-.
- Recognize KCN provides CN- nucleophile for substitution.
- Note standard condition requires ethanol solvent to dissolve KCN and favor SN2 over elimination or side reactions.
- Match options: only B specifies ethanol solvent with heat and KCN.
Why B is correct:
- Ethanol as solvent dissolves KCN fully, promoting clean SN2 displacement to yield the nitrile without hydrolysis.
Why the others are wrong:
- A: Lacks solvent specification; aqueous conditions lead to KCN hydrolysis, reducing CN- availability.
- C: Identical to A; no ethanol means incomplete reaction or side products like alcohols.
- D: Ethanol alone provides no CN- source, so no substitution occurs.
Final answer: B
Topic: Nitrogen compounds
Practice more A Levels Chemistry (9701) questions on mMCQ.me