A Levels Chemistry (9701)•9701/11/M/J/25
Explanation
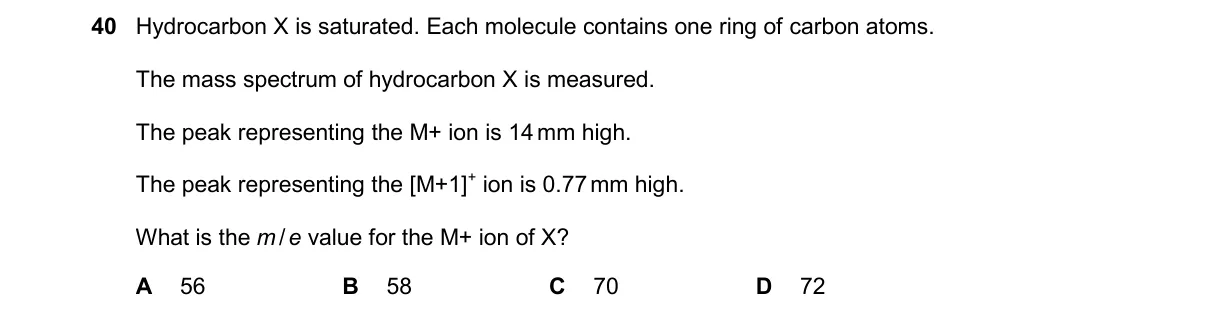
Molecular mass of C5H10 alkene Steps:
- Identify X as C4H10 from M+ = 58 (saturated alkane formula CnH2n+2: 12n + 2n + 2 = 58 gives n=4).
- Confirm fragmentation: [M-15]+ at 43 from loss of CH3• radical, typical for alkanes.
- For five-carbon homologue, saturated C5H12 mass = 12×5 + 12×1 = 72.
- Unsaturated version (one double bond) is C5H10, mass = 12×5 + 10×1 = 70.
Why C is correct:
- Alkene formula CnH2n yields 14n = 70 for n=5, per general hydrocarbon mass calculation.
Why the others are wrong:
- A. 56 m/z matches C4H8 (tetene, unsaturated with four carbons).
- B. 58 is M+ for given saturated C4H10.
- D. 72 m/z is for saturated C5H12.
Final answer: C
Topic: Analytical techniques
Practice more A Levels Chemistry (9701) questions on mMCQ.me