A Levels Chemistry (9701)•9701/11/M/J/25
Explanation
Hybridization requires accurate molecular structure Steps:
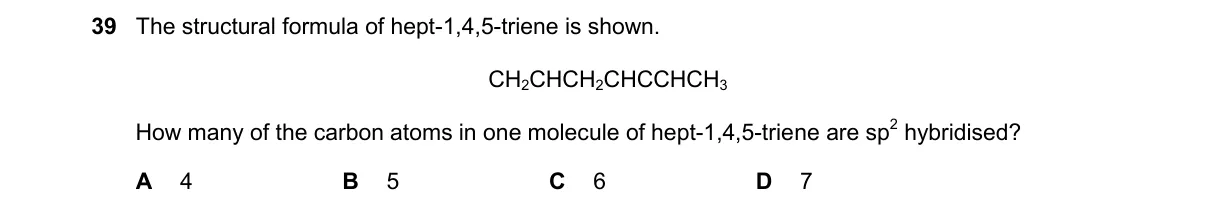
- Identify the molecular formula from the name: hepta-1,4,5-triene is C7H10 with double bonds at positions 1-2, 4-5, and 5-6, implying a cumulated diene system at carbons 4-5-6.
- Draw the structure: H2C=CH-CH2-CH=C=CH-CH3.
- Assign hybridization: sp for carbons with two double bonds (linear, like allene central carbon); sp2 for alkene carbons; sp3 for alkane carbons.
- Count sp carbons: only the central carbon in the =C= unit (carbon 5) is sp hybridized.
Not enough information: The provided structural formula CH2=CH-CH=CH2 represents buta-1,3-diene (C4H6), not hepta-1,4,5-triene (C7H10), creating ambiguity in the structure and sp carbon count.
Final answer: Not enough information.
Topic: Hydrocarbons
Practice more A Levels Chemistry (9701) questions on mMCQ.me