A Levels Chemistry (9701)•9701/11/M/J/25
Explanation
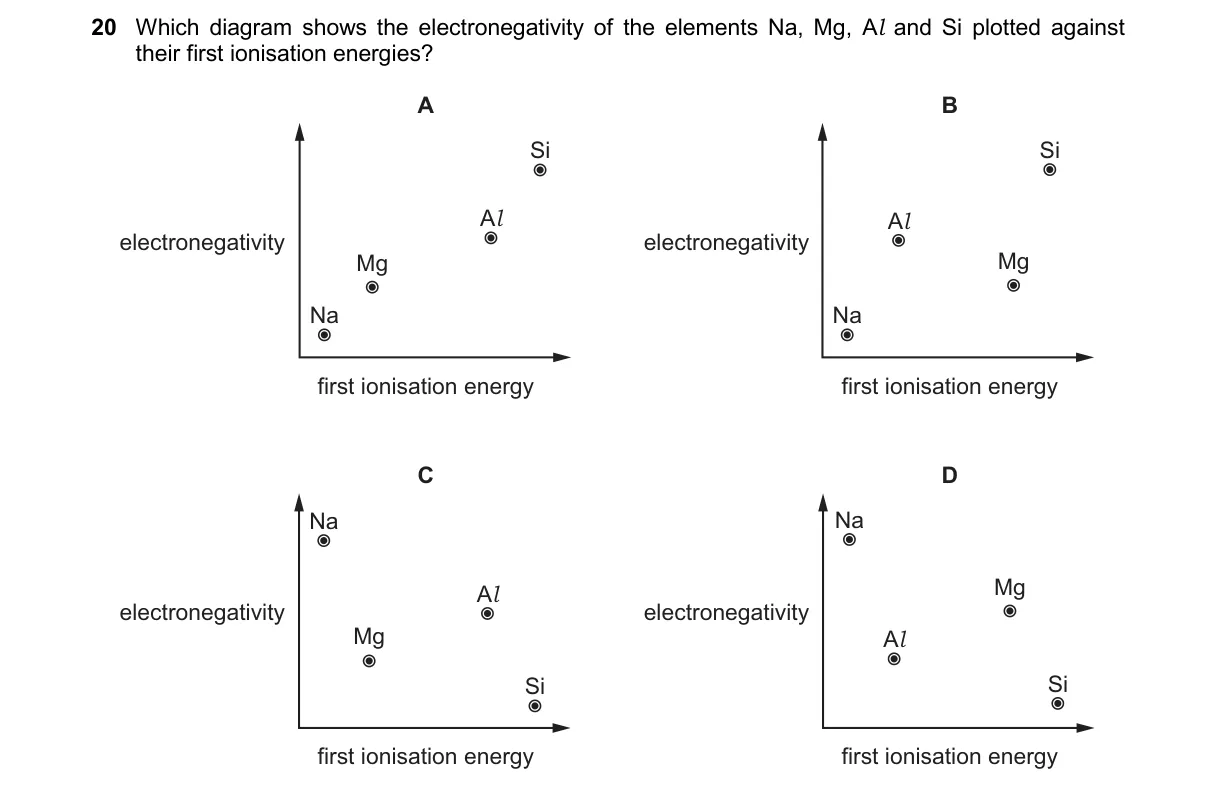
Trends in Period 3: Electronegativity vs. First Ionisation Energy
Steps:
- Identify elements in order: Na, Mg, Al, Si (period 3, left to right).
- Recall first ionisation energies (IE): Na (496 kJ/mol) < Al (578) < Mg (738) < Si (787), with dip at Al due to p-orbital removal.
- Recall electronegativities (EN, Pauling scale): Na (0.9) < Mg (1.2) < Al (1.5) < Si (1.8), steadily increasing across period.
- Plot EN (y-axis) vs. IE (x-axis): Points rise overall, but Al's IE dips below Mg's line while EN increases.
Why B is correct:
- B accurately plots the IE dip at Al against steady EN rise, matching periodic trends where EN increases with effective nuclear charge, unlike IE affected by subshell stability.
Why the others are wrong:
- A shows steady linear increase without Al's IE dip, ignoring p-subshell effect.
- C reverses axes or trends, plotting IE vs. EN incorrectly.
- D misorders elements or values, e.g., placing Si's high IE with low EN.
Final answer: B
Topic: The Periodic Table: chemical periodicity
Practice more A Levels Chemistry (9701) questions on mMCQ.me