A Levels Chemistry (9701)•9701/12/M/J/24
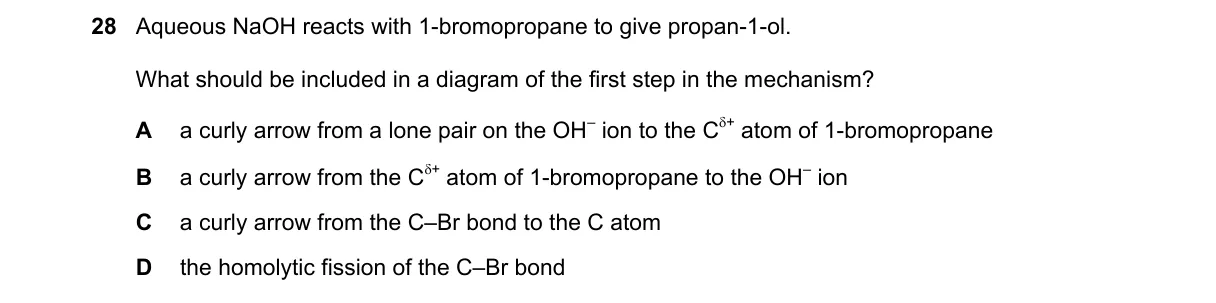
Explanation
SN2 Nucleophilic Attack in Substitution
Steps:
- Identify reaction type: Aqueous NaOH with primary alkyl halide (1-bromopropane) undergoes SN2 mechanism.
- Recall SN2 first step: Nucleophile (OH⁻) attacks carbon from lone pair, forming partial bond.
- Match to diagram: Curly arrow shows electron movement from OH⁻ lone pair to C atom.
- Eliminate mismatches: Other options depict incorrect electron flow or bond breaking.
Why A is correct:
- In SN2, the nucleophile's lone pair donates electrons to the electrophilic carbon, per the definition of nucleophilic substitution.
Why the others are wrong:
- B: Describes C-Br bond polarization but not the initial nucleophilic attack.
- C: Reverses electron flow; Br⁻ is leaving group, not donor.
- D: Indicates radical mechanism via equal electron split, irrelevant to ionic SN2.
Final answer: A
Topic: Halogen compounds
Practice more A Levels Chemistry (9701) questions on mMCQ.me