A Levels Chemistry (9701)•9701/11/M/J/24
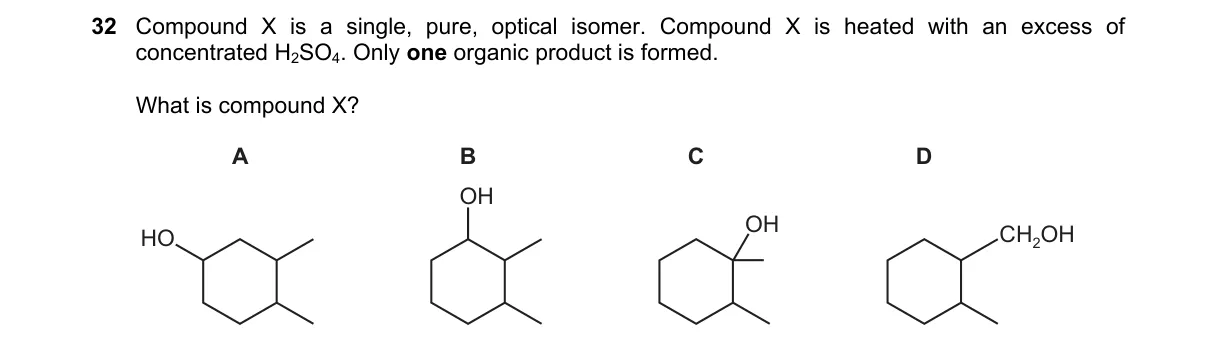
Explanation
Dehydration of chiral secondary alcohol to single alkene
Steps:
- X is a pure optical isomer, so it has a chiral center and is likely a secondary alcohol.
- Heating with excess H2SO4 triggers acid-catalyzed dehydration via E1, forming a planar carbocation and then an alkene.
- For only one organic product, the reaction must yield a single alkene with no constitutional or geometric isomers possible.
- Option D's structure allows carbocation formation leading exclusively to a terminal alkene like styrene, which is achiral and unique.
Why D is correct:
- D is 1-phenylethanol; dehydration forms the benzylic carbocation PhCH(+)-CH3, which loses the methyl proton to give only PhCH=CH2 (Zaitsev's rule favors this conjugated alkene).
Why the others are wrong:
- A: Aliphatic secondary alcohol like 2-butanol yields mixture of 1-butene, cis-/trans-2-butene.
- B: Unsymmetric alcohol like 3-pentanol gives cis-/trans-2-pentene isomers.
- C: Primary alcohol like 2-methyl-1-propanol dehydrates slowly to isobutene but may form ethers or multiple products under excess acid.
Final answer: D
Topic: Hydroxy compounds
Practice more A Levels Chemistry (9701) questions on mMCQ.me