A Levels Chemistry (9701)•9701/13/M/J/23
Explanation
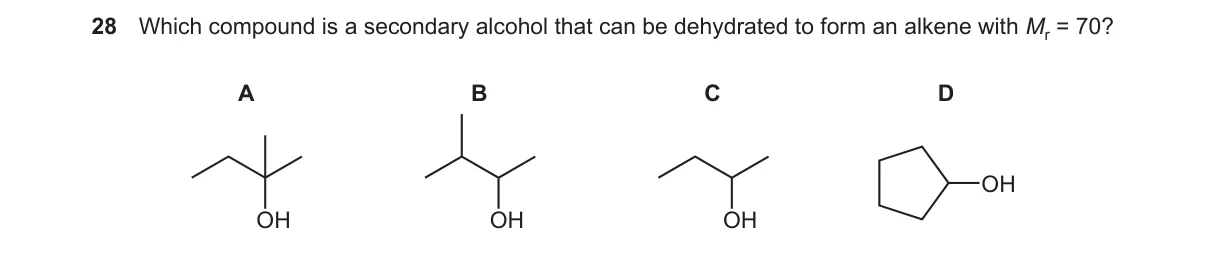
Secondary alcohol C5H12O dehydrates to C5H10 alkene (M+=70)
Steps:
- Secondary alcohol has OH on carbon bonded to two alkyl groups.
- Dehydration yields alkene by losing H2O (mass loss of 18).
- Alkene M+=70 corresponds to C5H10 (5×12 + 10×1 = 70), so alcohol is C5H12O (M+=88).
- Evaluate choices for secondary C5H12O structure.
Why B is correct:
- B is 3-pentanol (CH3CH2CH(OH)CH2CH3), a secondary alcohol (C5H12O) that dehydrates to pentene (C5H10, M+=70) per Zaitsev's rule.
Why the others are wrong:
- A: 1-Pentanol is primary (OH on terminal carbon).
- C: 2-Methyl-2-butanol is tertiary (OH on carbon with three alkyl groups).
- D: Cyclohexanol is C6H12O (M+=100), not C5H12O.
Final answer: B
Topic: Hydroxy compounds
Practice more A Levels Chemistry (9701) questions on mMCQ.me