A Levels Chemistry (9701)•9701/12/M/J/23
Explanation
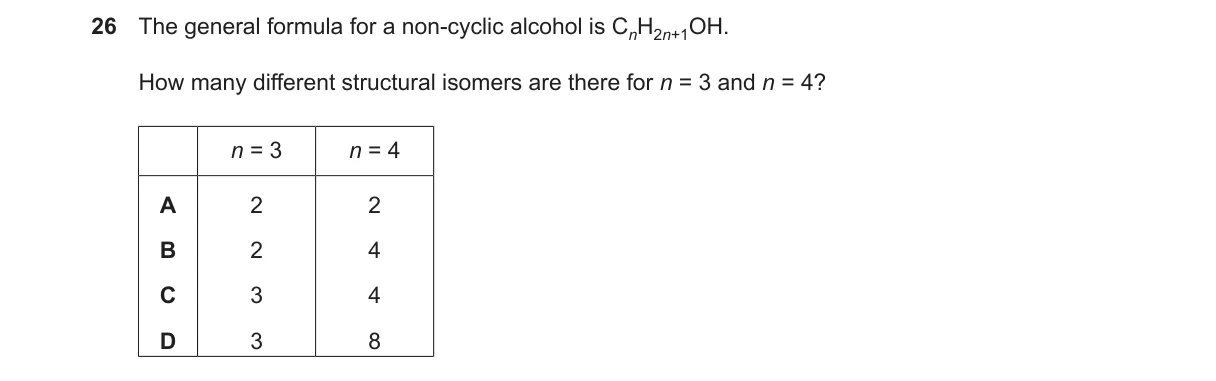
Counting structural isomers of non-cyclic alcohols
Steps:
- For n=3 (C₃H₈O): Straight chain allows OH on carbon 1 (1-propanol) or carbon 2 (2-propanol); no branches possible. Total: 2 isomers.
- For n=4 (C₄H₁₀O): Straight chain gives 1-butanol (OH on C1) and 2-butanol (OH on C2); branched chain (2-methylpropane) gives 2-methyl-1-propanol (primary OH) and 2-methyl-2-propanol (tertiary OH). Total: 4 isomers.
- Confirm non-cyclic: All are acyclic saturated alcohols fitting CₙH₂ₙ₊₂O.
Why B is correct:
- B gives 2 for n=3 and 4 for n=4, exactly matching the structural isomers from distinct carbon skeletons and OH positions.
Why the others are wrong:
- A: Lists 2 4, but overcounts or misapplies for n=4 by ignoring valid branched structures.
- C: 3 8 inflates n=3 count (only 2 exist) and vastly overcounts n=4 (only 4, not including ethers or cycles).
- D: Same as C, incorrect double-counting beyond alcohol isomers.
Final answer: B
Topic: Hydroxy compounds
Practice more A Levels Chemistry (9701) questions on mMCQ.me