A Levels Chemistry (9701)•9701/12/M/J/23
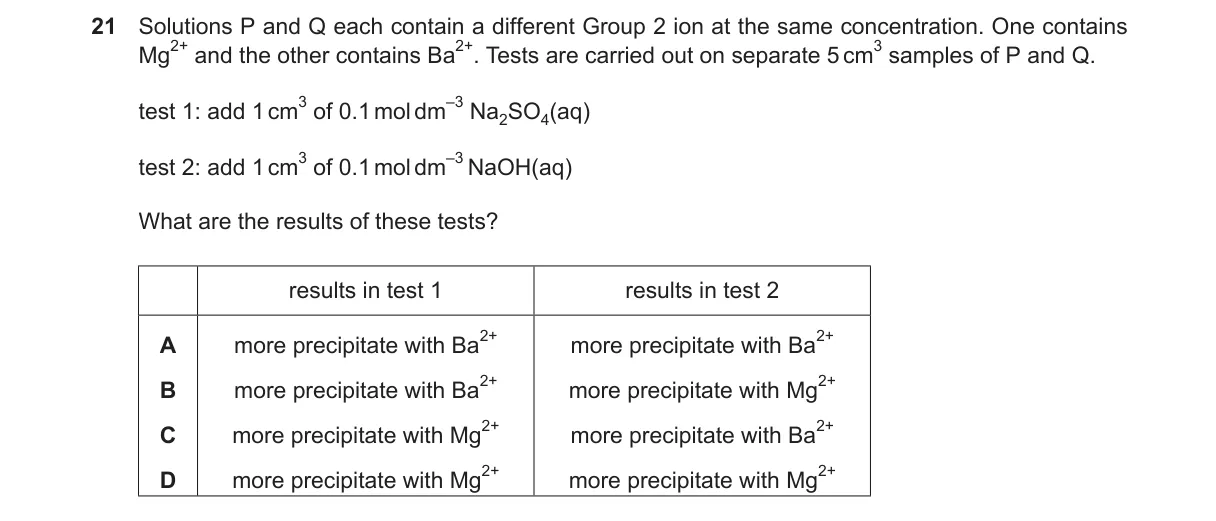
Explanation
Solubility trends in Group 2 compounds Steps:
- Test 1 adds SO₄²⁻; BaSO₄ has very low Ksp (1.1 × 10^{-10}), so precipitates heavily with Ba²⁺; MgSO₄ is soluble (high solubility), no precipitate with Mg²⁺.
- Test 2 adds OH⁻ to ~0.017 mol dm⁻³; Mg(OH)₂ has low Ksp (5.6 × 10^{-12}), so ion product exceeds Ksp and precipitates with Mg²⁺.
- Ba(OH)₂ has higher Ksp (~10^{-3}), ion product below Ksp at typical concentrations, so minimal/no precipitate with Ba²⁺.
- Results: more precipitate with Ba²⁺ in Test 1, more with Mg²⁺ in Test 2, matching option A.
Why A is correct:
- Matches solubility rules: sulfates become less soluble down Group 2 (Mg soluble, Ba insoluble); hydroxides become more soluble down Group 2 (Mg insoluble, Ba soluble).
Why the others are wrong:
- B: Incorrect for Test 2, as Ba(OH)₂ does not precipitate significantly.
- C: Incorrect for Test 1, as MgSO₄ does not precipitate.
- D: Reverses both tests, ignoring Group 2 solubility trends.
Final answer: A
Topic: Group 2
Practice more A Levels Chemistry (9701) questions on mMCQ.me