A Levels Chemistry (9701)•9701/12/M/J/22
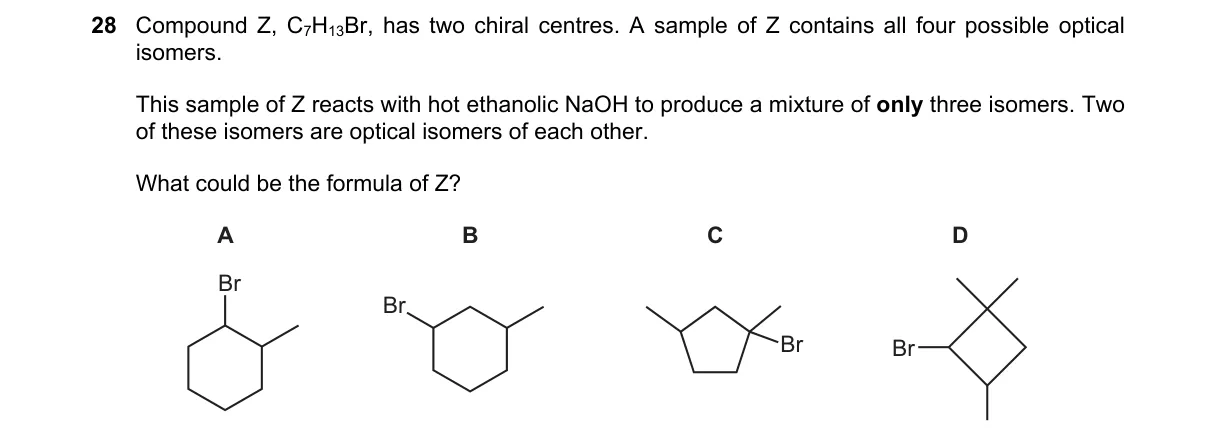
Explanation
Vicinal dibromide Z undergoes double dehydrohalogenation to three C4H4 isomers Steps:
- Hot ethanolic NaOH promotes double E2 elimination from C4H6Br2, yielding C4H4 products.
- Z's two chiral centers enable stereospecific paths to 1-buten-3-yne (achiral) and 1,2-butadiene (chiral allene).
- Elimination across the vicinal Br's in one direction gives 1-buten-3-yne; orthogonal path yields the enantiomeric 1,2-butadienes.
- Total: one achiral + enantiomeric pair = three isomers, with two optical.
Why A is correct:
- A's structure (e.g., 1,2-dibromo-2-methylcyclopropane) has two chiral centers and Br placement allowing exactly these elimination products per Zaitsev rule for alkynes/allenes.
Why the others are wrong:
- B: Yields four stereoisomers, exceeding three.
- C: No chiral centers, so no optical product pair.
- D: Produces only two achiral isomers.
Final answer: A
Topic: Halogen compounds
Practice more A Levels Chemistry (9701) questions on mMCQ.me