A Levels Chemistry (9701)•9701/11/M/J/22
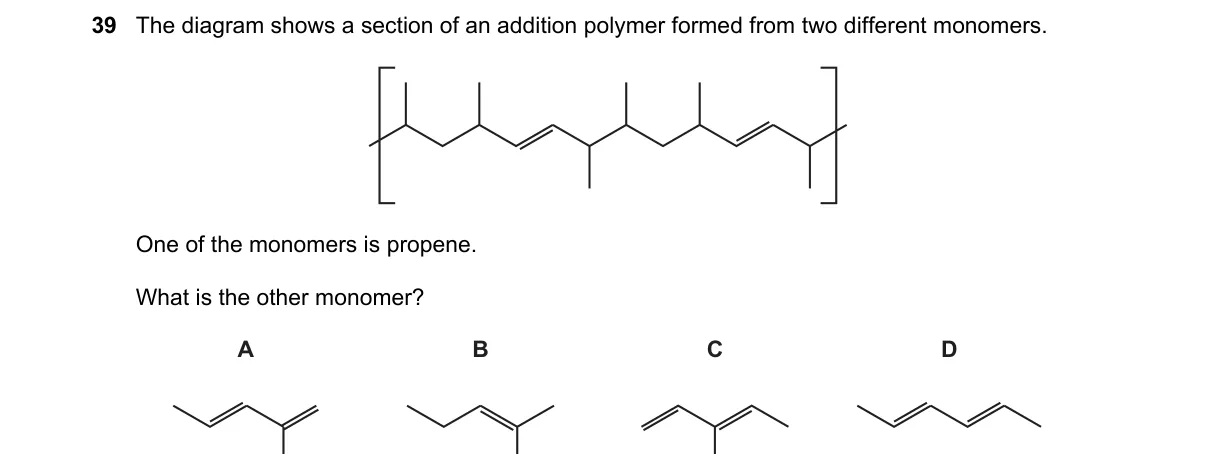
Explanation
Copolymer Structure Reveals Second Monomer
Steps:
- Analyze the polymer diagram for repeating units from two monomers.
- Confirm propene unit as -CH₂-CH(CH₃)- from CH₃CH=CH₂.
- Identify adjacent unit as -CH₂-CH₂-, indicating ethene (CH₂=CH₂) as the other monomer.
- Compare to choices; D matches ethene structure.
Why D is correct:
- Ethene (CH₂=CH₂) undergoes addition polymerization to form -CH₂-CH₂- units, directly matching the diagram's copolymer linkage with propene.
Why the others are wrong:
- A: Shows ethane (CH₃CH₃), a saturated alkane unable to form addition polymers.
- B: Depicts propene again, but the polymer uses two different monomers.
- C: Represents a branched alkene like butene, yielding mismatched -CH₂-CH(C₂H₅)- units.
Final answer: D
Topic: Polymerisation
Practice more A Levels Chemistry (9701) questions on mMCQ.me