A Levels Chemistry (9701)•9701/13/M/J/21
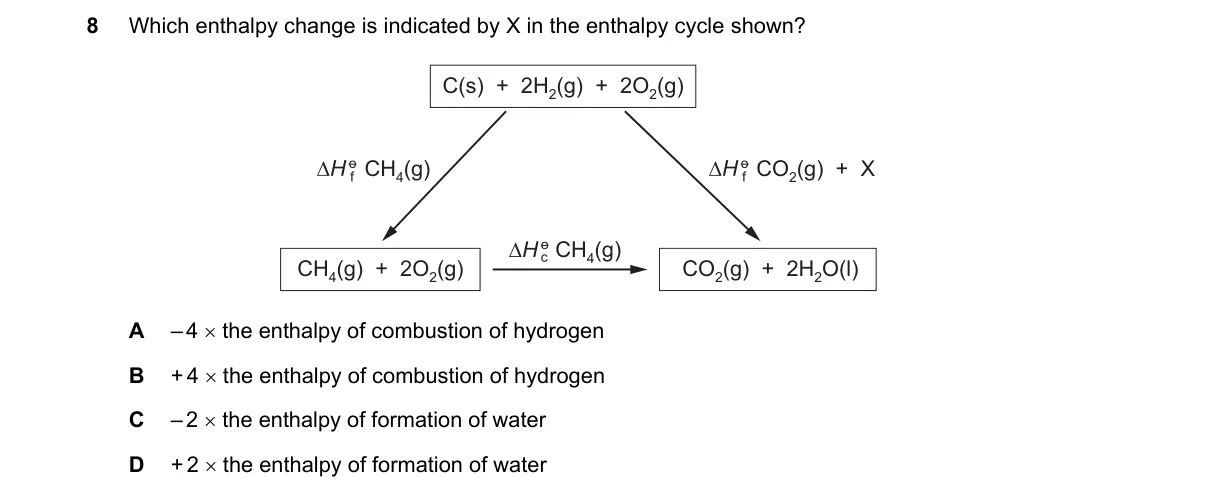
Explanation
Enthalpy cycle identifies formation enthalpy via Hess's law
Steps:
- Identify the path in the cycle where X represents the direct formation of water from elements.
- Apply Hess's law: total enthalpy change is path-independent.
- Trace reactants (H₂ + ½O₂) to product (H₂O) for the formation reaction.
- Confirm X matches ΔH_f for H₂O(l) or (g).
Why D is correct:
- Enthalpy of formation is defined as the enthalpy change when 1 mole of compound forms from elements in standard states, matching X's path.
Why the others are wrong:
- A: Combustion of H₂ forms water but involves excess oxygen, not formation.
- B: Involves methane, absent from water's cycle.
- C: Multiplies formation by 2, but X shows single mole formation.
Not enough information: Enthalpy cycle diagram not provided.
Final answer: D
Topic: Chemical energetics
Practice more A Levels Chemistry (9701) questions on mMCQ.me