A Levels Chemistry (9701)•9701/13/M/J/21
Explanation
Bond lengths follow bond order and atomic size trends
Steps:
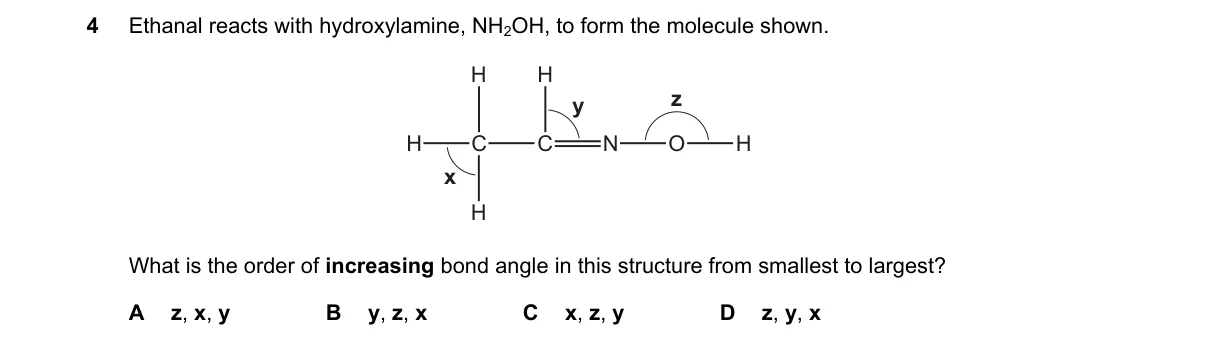
- Identify bonds in the oxime CH3CH=NOH: x labels C=N (double bond), y labels N-O (single bond), z labels C-C (single bond).
- Recall bond order rule: double bonds (C=N) are shorter than single bonds due to higher electron density.
- Compare single bonds: N-O (~1.46 Å) is shorter than C-C (~1.54 Å) because nitrogen and oxygen are smaller atoms than carbon.
- Order by increasing length: C=N (x) < N-O (y) < C-C (z).
Why A is correct:
- A (x y z) matches the trend where bond order decreases from double to single, and single bond length increases with atomic size.
Why the others are wrong:
- B: Places y (N-O) before x (C=N), but double bonds are always shorter than single bonds.
- C: Places z (C-C) before y (N-O), but N-O is shorter than C-C due to smaller atoms.
- D: Starts with y (N-O) as shortest, ignoring that C=N double bond is shorter.
Final answer: A
Topic: Carbonyl compounds
Practice more A Levels Chemistry (9701) questions on mMCQ.me