A Levels Chemistry (9701)•9701/13/M/J/21
Explanation
Iodoform and Tollens' Tests for Carbonyls
Steps:
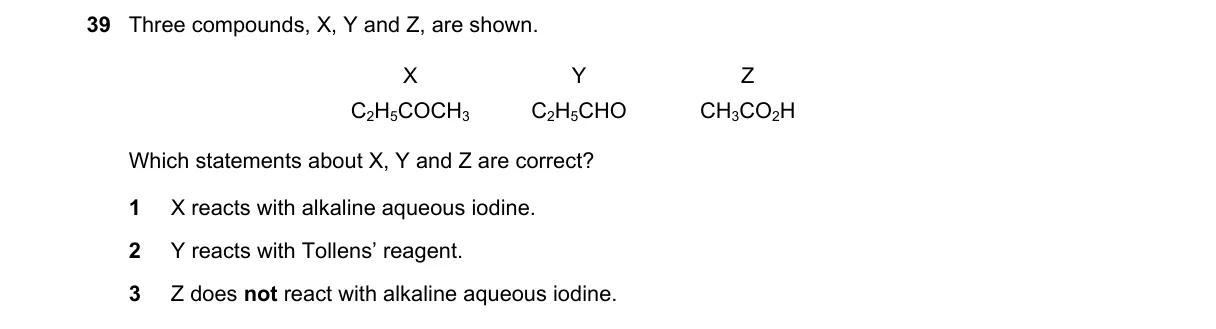
- Identify structures: X is acetaldehyde (CH₃CHO), Y is acetone (CH₃COCH₃), Z is propanal (CH₃CH₂CHO).
- Recall iodoform test (alkaline iodine): Positive for methyl ketones (CH₃CO-) and acetaldehyde; X and Y react, Z does not.
- Recall Tollens' reagent: Oxidizes aldehydes to carboxylic acids; ketones like Y do not react.
- Evaluate statements: 1 true (X gives iodoform), 2 false (Y inert to Tollens'), 3 true (Z no iodoform).
Why A is correct:
- A identifies statements 1 and 3 as true, matching iodoform reactivity for acetaldehyde (X) and non-methyl carbonyl (Z).
Why the others are wrong:
- B includes statement 2, but ketones do not reduce Tollens' reagent.
- C excludes statement 1, but acetaldehyde gives iodoform via CH₃CH(OH)- formation.
- D excludes statement 3, but propanal lacks CH₃CO- for iodoform.
Final answer: A
Topic: Carbonyl compounds
Practice more A Levels Chemistry (9701) questions on mMCQ.me