A Levels Chemistry (9701)•9701/13/M/J/21
Explanation
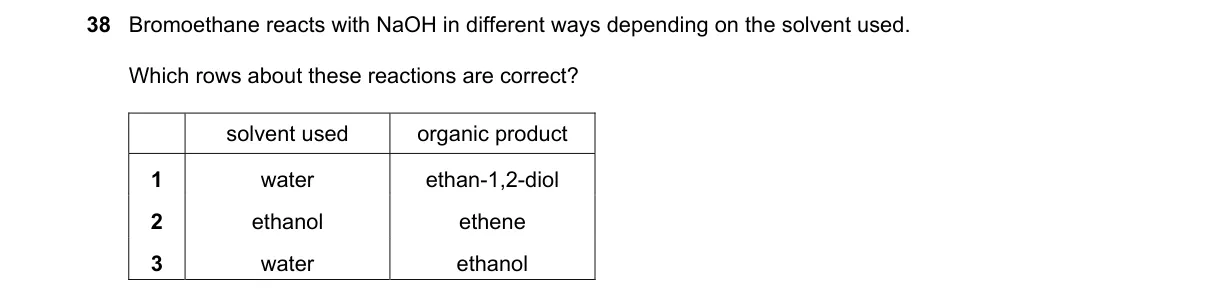
Solvent dictates substitution vs. elimination for bromoethane with NaOH
Steps:
- Bromoethane (CH₃CH₂Br) reacts with NaOH via SN2 in protic water, yielding substitution product ethanol (CH₃CH₂OH).
- In ethanolic NaOH, the less polar solvent promotes E2 elimination, yielding ethene (CH₂=CH₂).
- Evaluate row 1: ethanolic solvent should give ethene, not ethane-1,2-diol (HOCH₂CH₂OH), so incorrect.
- Evaluate rows 2 and 3: aqueous solvent favors substitution to ethanol, not elimination to ethene, so row 2 correct, row 3 incorrect.
Why C is correct:
- C identifies only row 2 as accurate, aligning with the rule that aqueous NaOH drives nucleophilic substitution per SN2 mechanism for primary alkyl halides.
Why the others are wrong:
- A includes row 1, but ethanolic NaOH yields ethene via E2, not diol.
- B includes row 1, ignoring elimination in ethanol.
- D includes row 3, but water favors substitution, not E2 elimination.
Final answer: C
Topic: Halogen compounds
Practice more A Levels Chemistry (9701) questions on mMCQ.me