A Levels Chemistry (9701)•9701/13/M/J/21
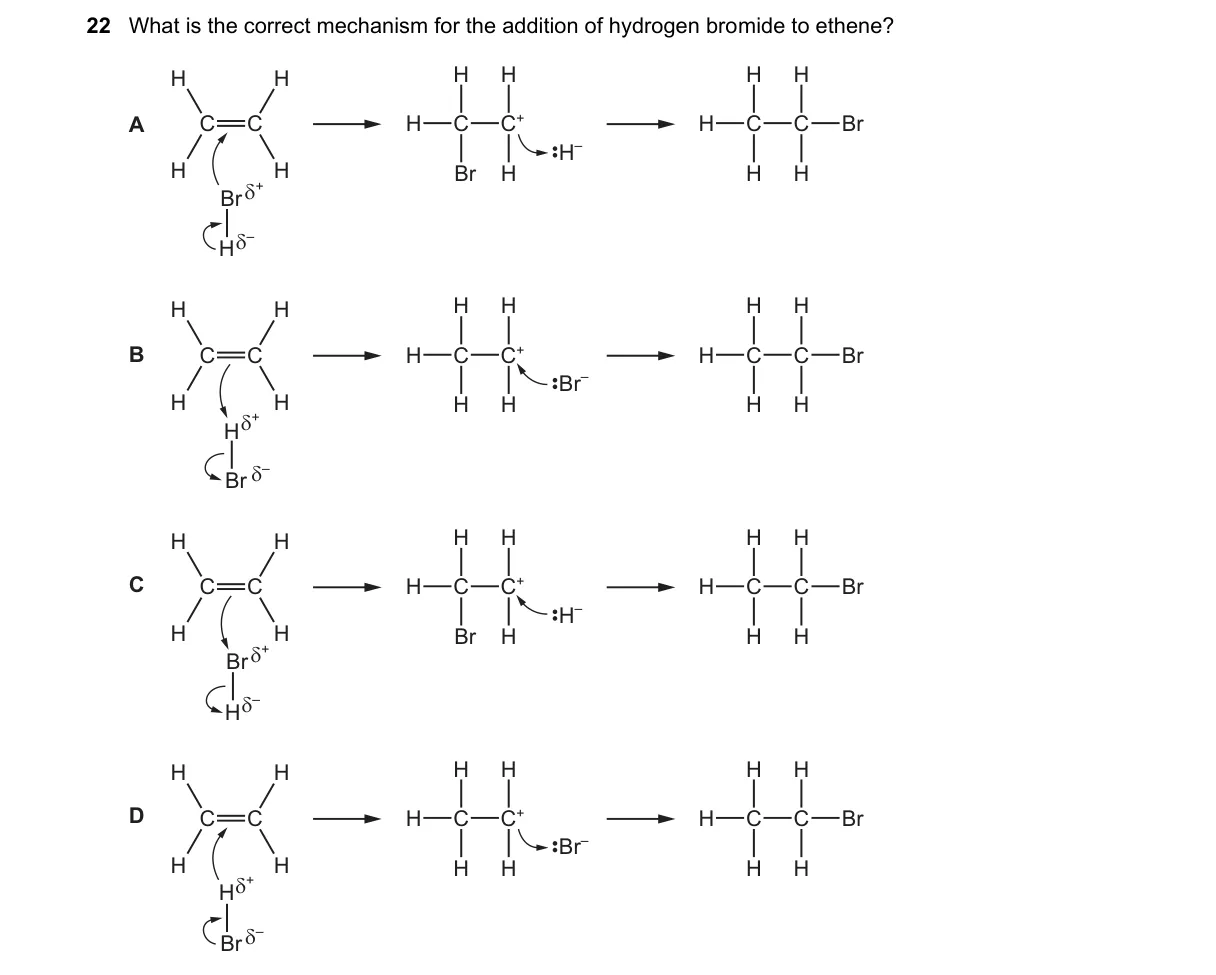
Explanation
Electrophilic addition mechanism for HBr to ethene
Steps:
- π bond of ethene attacks Hδ+ from HBr, heterolytically cleaving H-Br bond.
- Forms ethyl carbocation intermediate +CH2-CH3 (or H2C-CH3 with + on first C) and free Brδ-.
- Brδ- nucleophilically attacks positively charged carbon.
- Yields CH3-CH2Br as product, following Markovnikov orientation (symmetric here).
Why B is correct:
- Depicts carbocation intermediate H2C-CH2+ with separate Brδ-, matching H+ addition first per electrophilic addition rule.
Why the others are wrong:
- A: Intermediate H3C-CHBr+ implies Br+ electrophile, violating HBr polarity.
- C: Labels Brδ+, contradicting HBr's partial charges (Hδ+, Brδ-).
- D: Intermediate H2C-CHBr+ wrongly attaches Brδ- early, suggesting nucleophilic addition.
Final answer: B
Topic: Hydrocarbons
Practice more A Levels Chemistry (9701) questions on mMCQ.me