A Levels Chemistry (9701)•9701/12/M/J/21
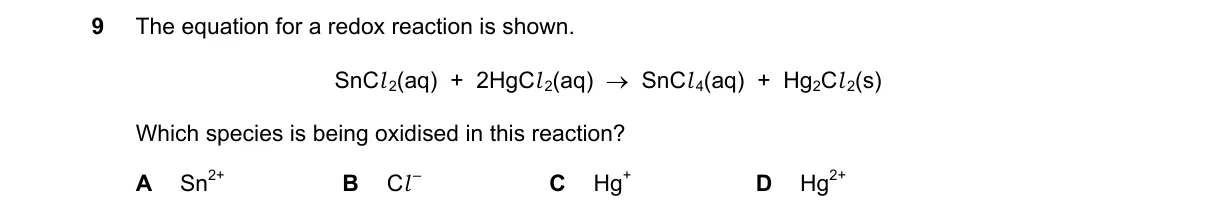
Explanation
Oxidation state increase identifies the oxidized species
Steps:
- Assign oxidation states: In SnCl₂, Sn is +2; in SnCl₄, Sn is +4. In HgCl₂, Hg is +2; in Hg₂Cl₂, each Hg is +1. Cl is -1 throughout.
- Compare changes: Sn oxidation state rises from +2 to +4 (loss of electrons).
- Hg oxidation state falls from +2 to +1 (gain of electrons).
- Cl shows no change, so Sn is oxidized.
Why A is correct:
- Sn²⁺ increases oxidation state from +2 to +4, losing two electrons per the definition of oxidation.
Why the others are wrong:
- B. Cl⁻ maintains -1 oxidation state in all compounds.
- C. Hg²⁺ decreases from +2 to +1 in Hg₂²⁺, indicating reduction.
- D. Hg₂²⁺ forms as the reduced product, not undergoing oxidation.
Final answer: A
Topic: Electrochemistry
Practice more A Levels Chemistry (9701) questions on mMCQ.me