A Levels Chemistry (9701)•9701/12/M/J/21
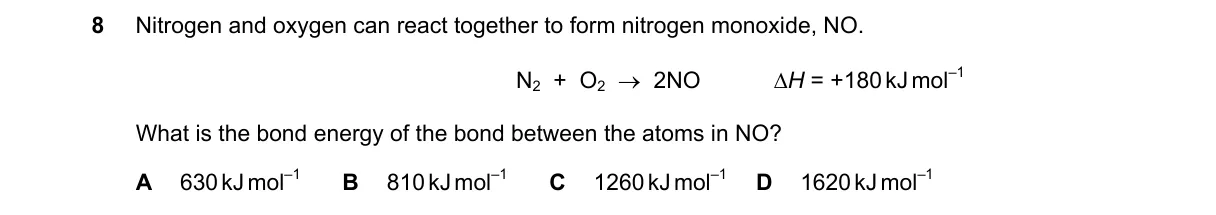
Explanation
Bond Energy Calculation from Reaction Enthalpy
Steps:
- Bonds broken: N≡N (941 kJ mol⁻¹) and O=O (498 kJ mol⁻¹), total 1439 kJ mol⁻¹.
- Bonds formed: 2 N=O bonds, releasing 2 × BE(NO).
- ΔH = energy absorbed to break bonds - energy released to form bonds = +180 kJ mol⁻¹.
- 1439 - 2 × BE(NO) = 180, so 2 × BE(NO) = 1259 kJ mol⁻¹.
- BE(NO) = 1259 / 2 = 630 kJ mol⁻¹.
Why A is correct:
- Matches the value from the bond energy equation using standard N≡N and O=O values with given ΔH.
Why the others are wrong:
- B: Gives ΔH ≈ -181 kJ mol⁻¹ (exothermic), contradicting +180 kJ mol⁻¹.
- C: Equals total energy for 2 N=O bonds, not single bond energy.
- D: Gives ΔH ≈ -181 kJ mol⁻¹ (exothermic), contradicting +180 kJ mol⁻¹.
Final answer: A
Topic: Chemical energetics
Practice more A Levels Chemistry (9701) questions on mMCQ.me