A Levels Chemistry (9701)•9701/12/M/J/21
Explanation
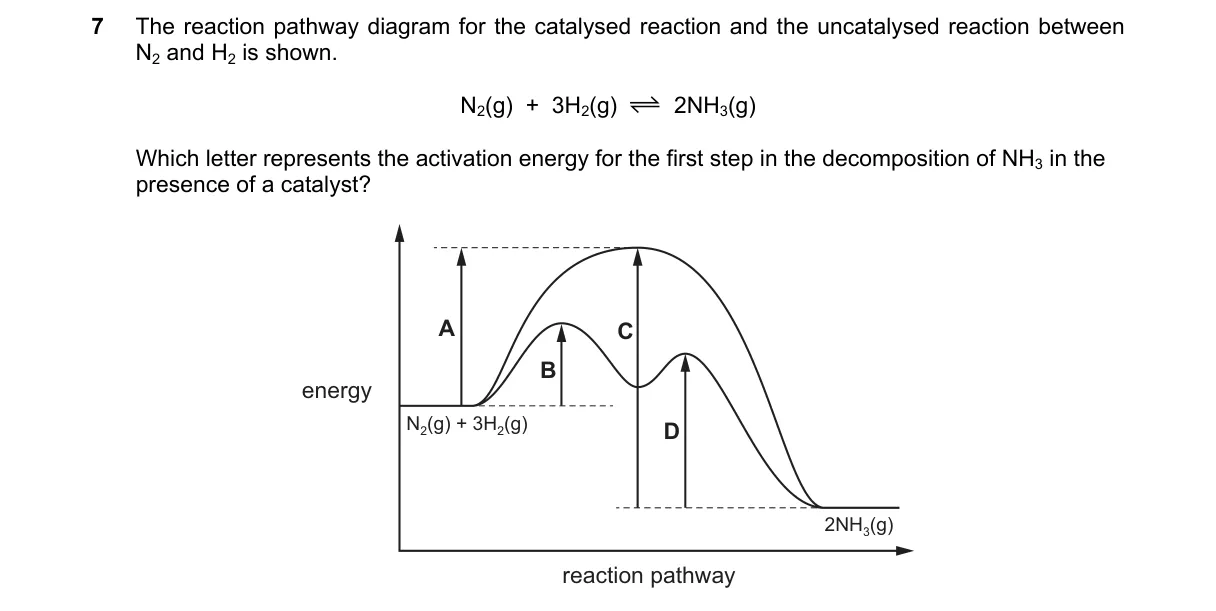
Activation Energy in Catalytic Decomposition
Steps:
- Examine the energy diagram for the catalyzed decomposition of NH3, showing stepwise energy changes.
- Identify the reactants' energy level as the starting point.
- Locate the highest energy point (transition state) in the first reaction step after the catalyst lowers the barrier.
- Measure the energy difference from reactants to this first transition state; this is the activation energy for the first step.
Why D is correct:
- D labels the vertical distance from reactant energy to the first transition state peak, matching the definition of activation energy as the minimum energy required to initiate the reaction (Arrhenius equation: rate constant depends on e^(-Ea/RT)).
Why the others are wrong:
- A represents overall reaction energy change, not the first step's barrier.
- B indicates the second step's activation energy, beyond the first step.
- C shows the catalyzed overall activation energy, not specific to the first step.
Final answer: D
Topic: Reaction kinetics
Practice more A Levels Chemistry (9701) questions on mMCQ.me