A Levels Chemistry (9701)•9701/12/M/J/21
Explanation
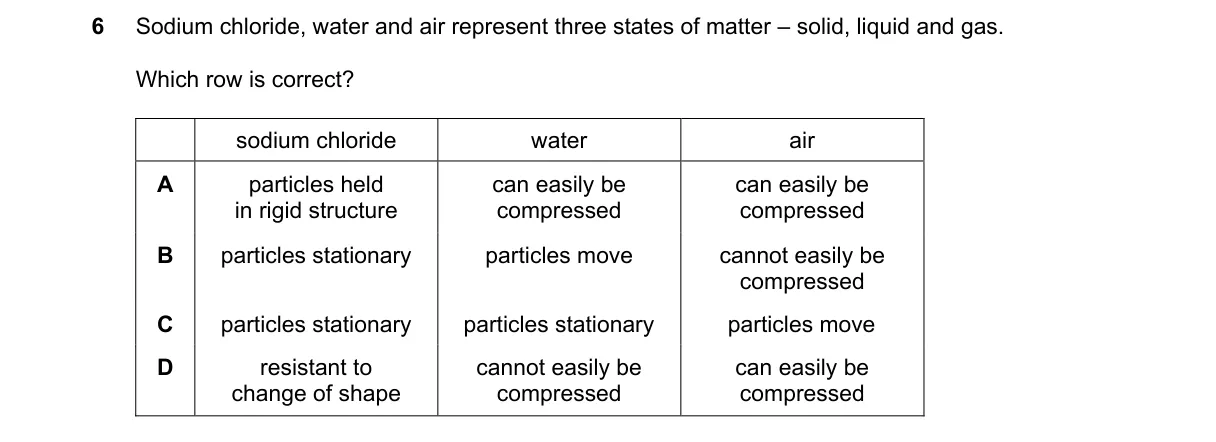
Compressibility distinguishes states of matter
Steps:
- Identify states: sodium chloride as solid, water as liquid, air as gas.
- Recall key property: solids and liquids resist compression due to tightly packed particles; gases compress easily due to spaced-out particles.
- Evaluate options: match sequence to solid (incompressible), liquid (incompressible), gas (compressible).
- Confirm D fits perfectly.
Why D is correct:
- Incompressibility defines solids and liquids by close particle packing (kinetic molecular theory), while gases compress via large intermolecular spaces.
Why the others are wrong:
- A: Incorrectly states liquids and gases compress easily; liquids do not.
- B: Falsely claims solids have stationary particles (they vibrate) and gases incompressible.
- C: Ignores compressibility; also wrongly says liquid particles stationary (they slide past each other).
Final answer: D
Topic: States of matter
Practice more A Levels Chemistry (9701) questions on mMCQ.me