A Levels Chemistry (9701)•9701/12/M/J/21
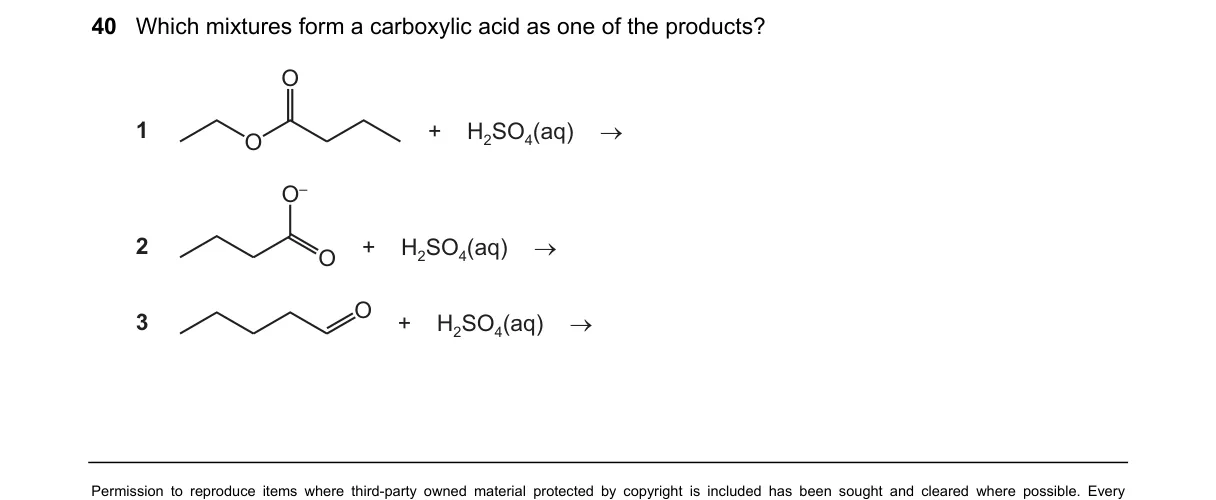
Explanation
Acid-catalyzed reactions of organic compounds with H2SO4(aq) Steps:
- Identify the functional groups: A and B are ketones, C is a primary alcohol.
- Recall reactions with dilute H2SO4(aq): Ketones form no products; primary alcohols dehydrate only under concentrated/hot conditions to alkenes, not carboxylic acids.
- Check for oxidation: H2SO4(aq) alone lacks an oxidant (e.g., no Cr2O7^2-), so no carboxylic acid from alcohol.
- Note missing details: No halogens for haloform on ketones; D option absent.
Why B is correct:
- Not enough information; no clear reaction produces carboxylic acid from any option with just H2SO4(aq).
Why the others are wrong:
- A: Ketone unchanged; no carboxylic acid.
- C: Alcohol forms no carboxylic acid without oxidant.
Final answer: Not enough information.
Topic: Carboxylic acids and derivatives
Practice more A Levels Chemistry (9701) questions on mMCQ.me