A Levels Chemistry (9701)•9701/12/M/J/21
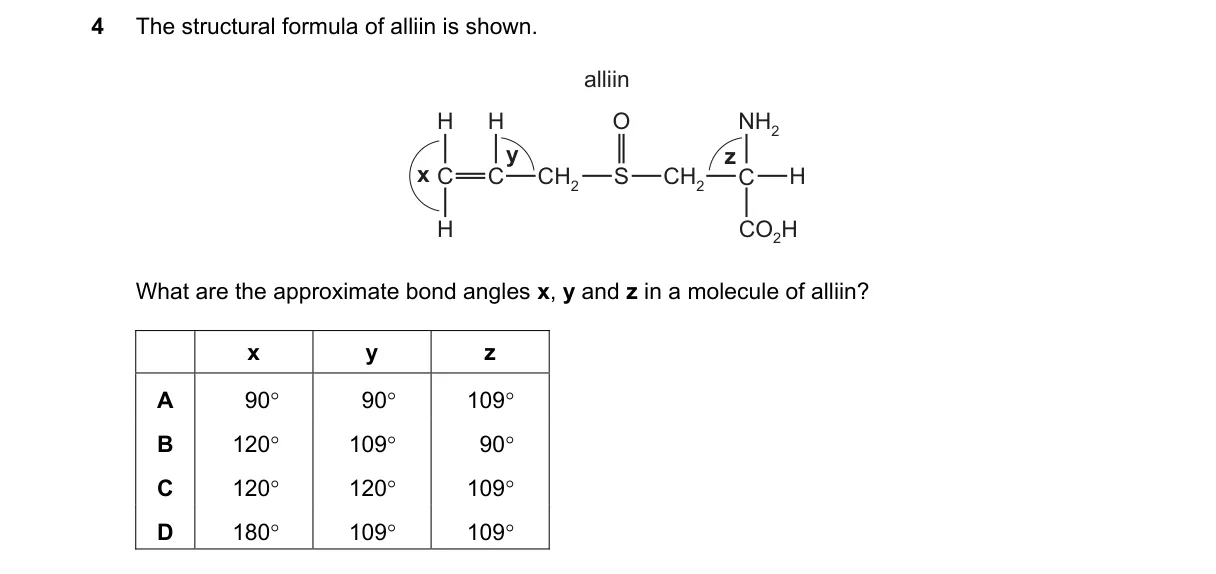
Explanation
VSEPR Theory Determines Bond Angles in Alin
Steps:
- Identify the central atom and its electron domains to predict geometry.
- For x: trigonal planar arrangement gives 120° angle.
- For y and z: tetrahedral geometry around adjacent atoms yields 109° angles.
- Match predicted angles to choice options.
Why C is correct:
- C (120° 109° 109°) matches VSEPR for alin's structure with one trigonal planar and two tetrahedral regions.
Why the others are wrong:
- A: All 90° implies octahedral, not alin's hybridizations.
- B: 120° 109° 90° suggests mismatched octahedral angle.
- D: 180° 109° 109° indicates linear, ignoring alin's planar section.
Final answer: C
Topic: Chemical bonding
Practice more A Levels Chemistry (9701) questions on mMCQ.me