A Levels Chemistry (9701)•9701/12/M/J/21
Explanation
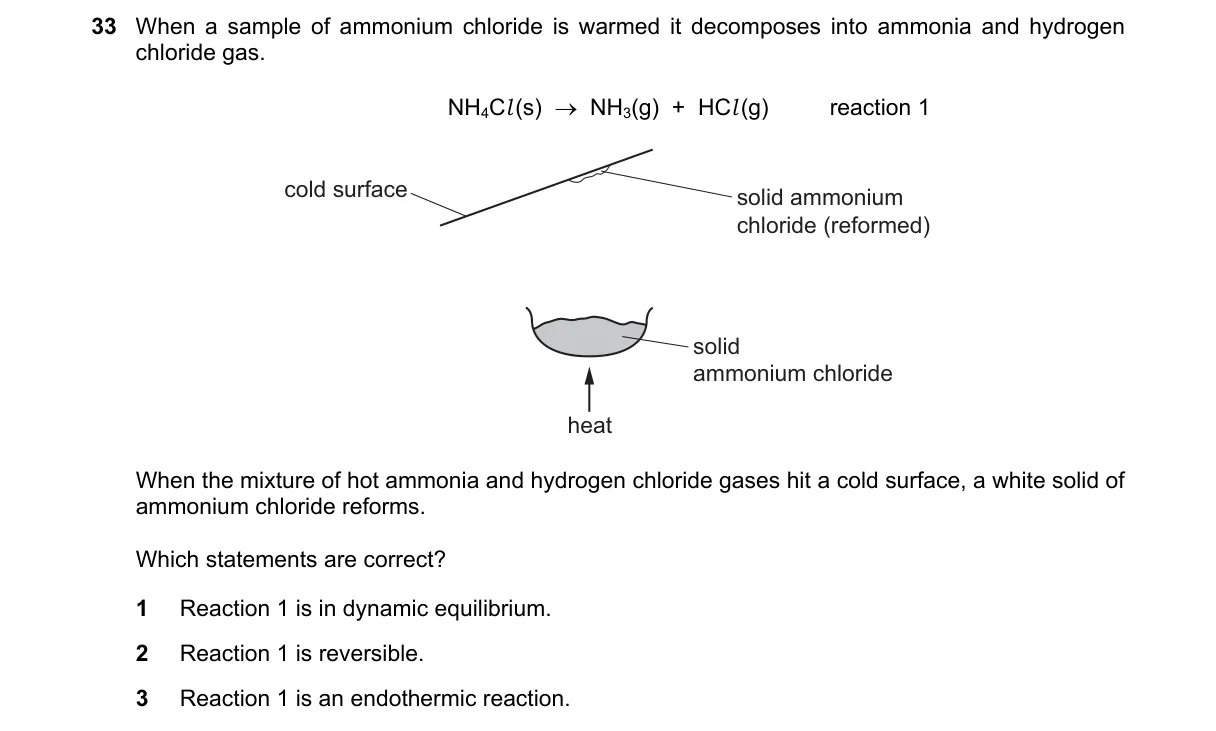
Reversible decomposition of ammonium chloride
Steps:
- Observe the forward reaction: warming NH4Cl(s) produces NH3(g) and HCl(g), indicating heat drives decomposition.
- Note the reverse: gases on cold surface reform NH4Cl(s), showing reversibility.
- Infer equilibrium: in a closed system, forward and reverse rates equalize dynamically.
- Determine thermodynamics: heat input for forward suggests endothermicity.
Why C is correct:
- Not enough information: Choices C and D are not provided, preventing evaluation.
Why the others are wrong:
- A: Dynamic equilibrium requires closed system with equal forward/reverse rates; open warming favors forward only.
- B: Reaction is endothermic (heat absorbed for decomposition), but statement lacks context for full assessment.
Final answer: Not enough information.
Topic: Equilibria
Practice more A Levels Chemistry (9701) questions on mMCQ.me