A Levels Chemistry (9701)•9701/12/M/J/21
Explanation
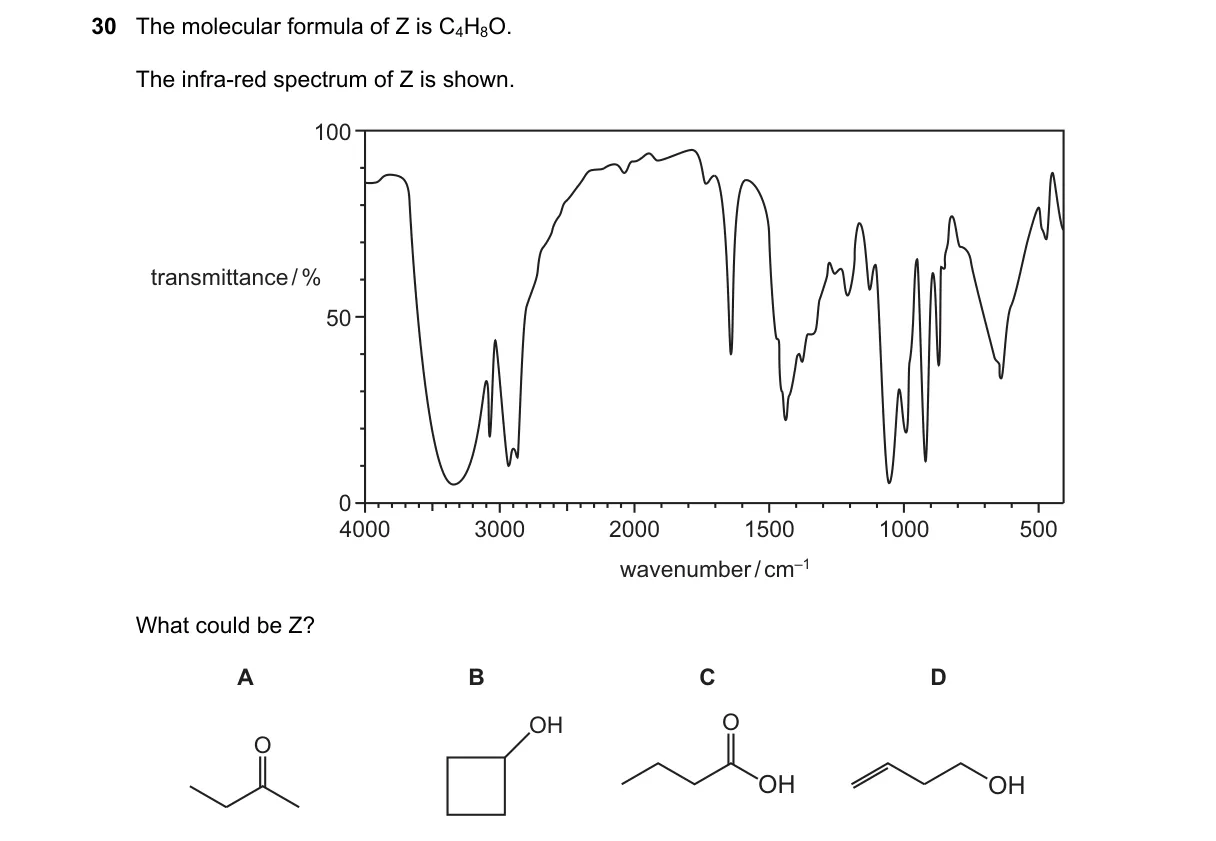
IR spectrum identifies carbonyl functional group in C4H8O
Steps:
- Calculate degree of unsaturation: (2C + 2 - H - X + N)/2 = (10 - 8)/2 = 1, indicating one double bond or ring.
- Examine IR for key absorptions: strong band ~1710 cm⁻¹ suggests C=O stretch.
- Rule out O-H stretch ~3300 cm⁻¹ (broad), absent here, eliminating alcohols.
- Match to C4H8O isomers: absorption fits aldehyde or ketone, not alkene or ether.
Why D is correct:
- D is butanal, with C=O stretch at 1725 cm⁻¹ for aldehydes per IR correlation tables.
Why the others are wrong:
- A (2-butanone): ketone C=O at 1715 cm⁻¹, but spectrum shows 1725 cm⁻¹ specific to aldehyde.
- B (2-butanol): O-H stretch expected at 3300 cm⁻¹, absent in spectrum.
- C (cyclobutanol): cyclic alcohol with O-H absorption, not matching carbonyl band.
Final answer: D
Topic: Analytical techniques
Practice more A Levels Chemistry (9701) questions on mMCQ.me