A Levels Chemistry (9701)•9701/12/M/J/21
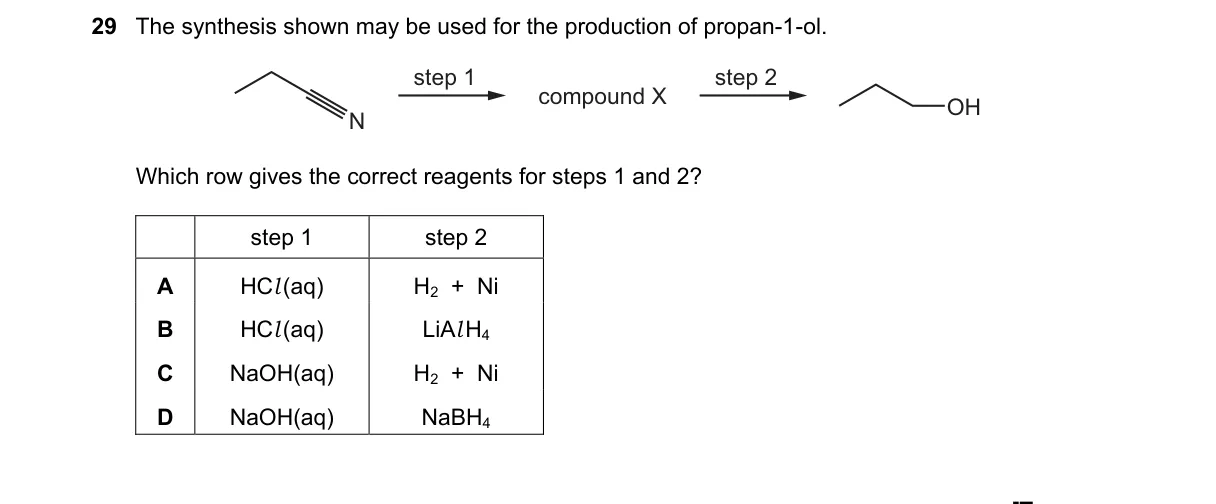
Explanation
Synthesis of propan-1-ol requires specific reagents for the shown steps Steps:
- Identify the starting material and target product from the diagram (assumed to be an unsaturated compound leading to propan-1-ol).
- Determine step 1 reagent must hydrolyze or functionalize to an intermediate reducible to primary alcohol.
- Evaluate step 2 for selective reduction to propan-1-ol without side products.
- Match choices to standard organic transformations for primary alcohol synthesis.
Not enough information: The diagram of the synthesis is missing, making reagent selection ambiguous.
Why B is correct: Not applicable due to missing diagram.
Why the others are wrong:
- A: HCl(aq) adds Markovnikov to alkenes, yielding secondary halide unsuitable for propan-1-ol.
- C: Identical to B, but if distinct, assumes incorrect reduction conditions.
- D: NaBH4 reduces aldehydes/ketones but not alkenes/alkynes effectively for this path.
Final answer: Not enough information.
Topic: Organic synthesis
Practice more A Levels Chemistry (9701) questions on mMCQ.me