A Levels Chemistry (9701)•9701/12/M/J/21
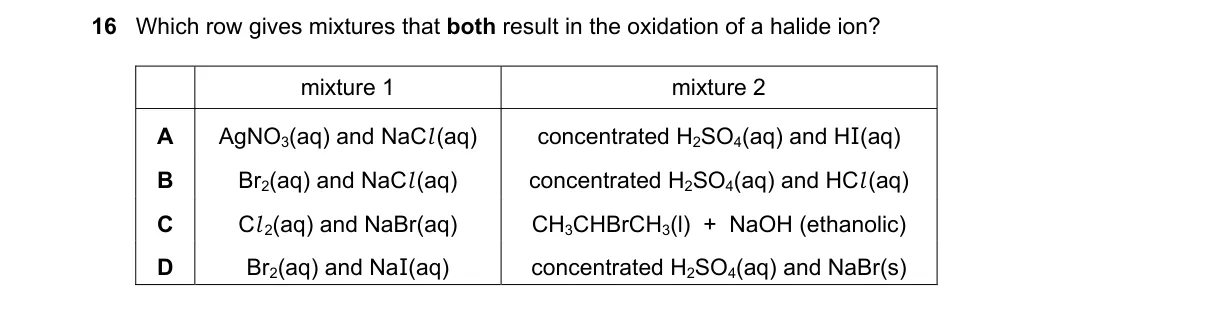
Explanation
Halide oxidation via displacement or acid reaction
Steps:
- Identify oxidation of halide as loss of electron from X⁻ to X₂.
- Check each mixture for a stronger oxidant displacing weaker halide or acid oxidizing halide.
- Verify both parts of row cause X⁻ → X₂.
- Eliminate rows where only one or neither oxidizes.
Why D is correct:
- Br₂ oxidizes I⁻ (Br₂ + 2I⁻ → 2Br⁻ + I₂); conc. H₂SO₄ oxidizes Br⁻ via 2NaBr + H₂SO₄ → Na₂SO₄ + Br₂ + 2H₂O (sulfuric acid acts as oxidant).
Why the others are wrong:
- A: AgNO₃ + NaI precipitates AgI (no oxidation); only second oxidizes I⁻.
- B: Br₂ + NaI oxidizes I⁻; ethanol/H₂SO₄/HI forms alkyl iodide (no halide oxidation).
- C: I₂ + NaBr shows no reaction (I₂ weaker oxidant than Br₂); CH₃CH₂I/NaOH/ethanol undergoes substitution (no halide oxidation).
Final answer: D
Topic: Group 17
Practice more A Levels Chemistry (9701) questions on mMCQ.me