A Levels Chemistry (9701)•9701/11/M/J/21
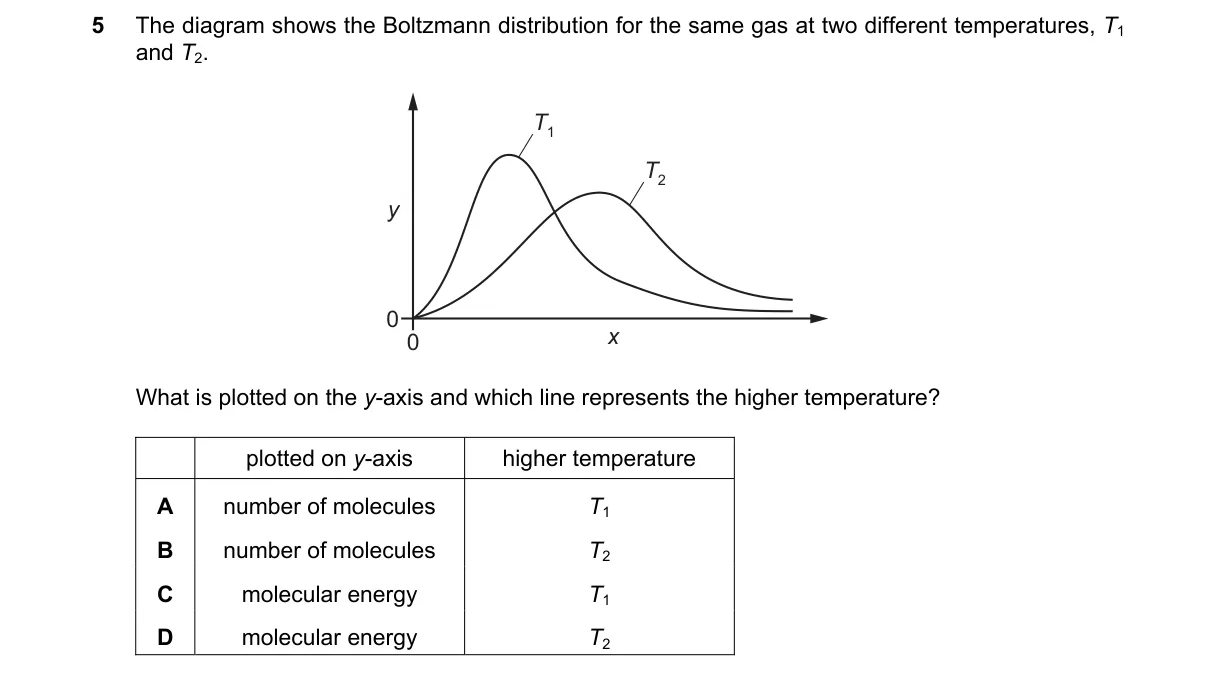
Explanation
Maxwell-Boltzmann Distribution Axes and Temperature
Steps:
- Examine the graph: y-axis typically plots the number of molecules at each kinetic energy level.
- Identify x-axis: it represents molecular kinetic energy.
- Compare curves: the higher-temperature line peaks at higher energy and has a lower maximum height due to broader spread.
- Determine labels: T1 curve shows this shift, indicating higher temperature.
Why B is correct:
- In kinetic theory, the y-axis is number of molecules (fraction with energy E), and T1 represents higher temperature as its curve shifts right per the Boltzmann factor e^(-E/kT), favoring higher energies at elevated T.
Why the others are wrong:
- A: T2 is lower temperature, with peak at lower energy.
- C: Y-axis is number of molecules, not energy; T2 is lower temperature.
- D: Y-axis is number of molecules, not energy; T1 is higher temperature.
Final answer: B
Topic: Reaction kinetics
Practice more A Levels Chemistry (9701) questions on mMCQ.me