A Levels Chemistry (9701)•9701/11/M/J/21
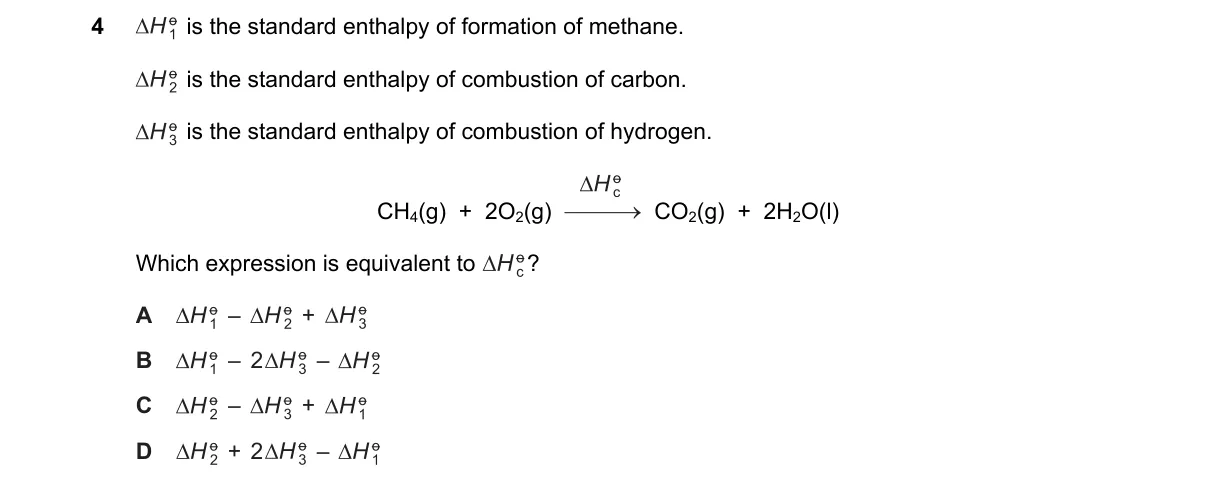
Explanation
Hess's Law Application to Enthalpy Relationships
Steps:
- Write the formation reaction reversed: CH₄(g) → C(s) + 2H₂(g), ΔH = -ΔH_f.
- Add carbon combustion: C(s) + O₂(g) → CO₂(g), ΔH = ΔH_c.
- Add hydrogen combustion (for 2 moles H₂): 2H₂(g) + O₂(g) → 2H₂O(l), ΔH = ΔH.
- Combine: CH₄(g) + 2O₂(g) → CO₂(g) + 2H₂O(l), ΔH = -ΔH_f + ΔH_c + ΔH.
Why D is correct:
- Hess's law states the total enthalpy change equals the sum of steps, so the reaction's ΔH = ΔH_c + ΔH - ΔH_f, matching D.
Why the others are wrong:
- A: Subtracts ΔH_c and ΔH from ΔH_f, reversing the combustion directions.
- B: Subtracts 2ΔH and ΔH_c from ΔH_f, incorrectly doubling hydrogen without adding carbon combustion.
- C: Adds 2ΔH instead of ΔH, assuming ΔH is per mole H₂ rather than for 2 moles.
Final answer: D
Topic: Chemical energetics
Practice more A Levels Chemistry (9701) questions on mMCQ.me