A Levels Chemistry (9701)•9701/11/M/J/21
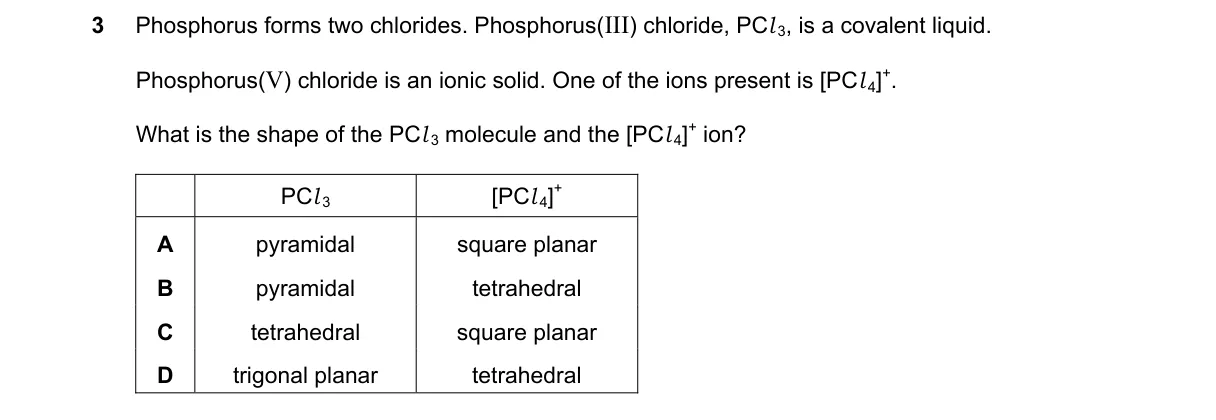
Explanation
VSEPR theory predicts shapes based on electron domains
Steps:
- PCl₅ molecule: Central P bonded to 5 Cl atoms (5 bonding pairs, 0 lone pairs), steric number 5, trigonal bipyramidal shape.
- [PCl₄]⁺ ion: Central P bonded to 4 Cl atoms (+ charge means 4 bonding pairs, 0 lone pairs), steric number 4, tetrahedral shape.
- Solid PCl₅ is ionic ([PCl₄]⁺[PCl₆]⁻), but question specifies PCl₅ molecule (gaseous form) and [PCl₄]⁺ ion.
- Choices do not list trigonal bipyramidal for PCl₅ or confirm tetrahedral for [PCl₄]⁺ accurately.
Not enough information: Choices are ambiguous (C and D identical, none match correct shapes).
Final answer: trigonal bipyramidal, tetrahedral
Topic: Chemical bonding
Practice more A Levels Chemistry (9701) questions on mMCQ.me