A Levels Chemistry (9701)•9701/11/M/J/21
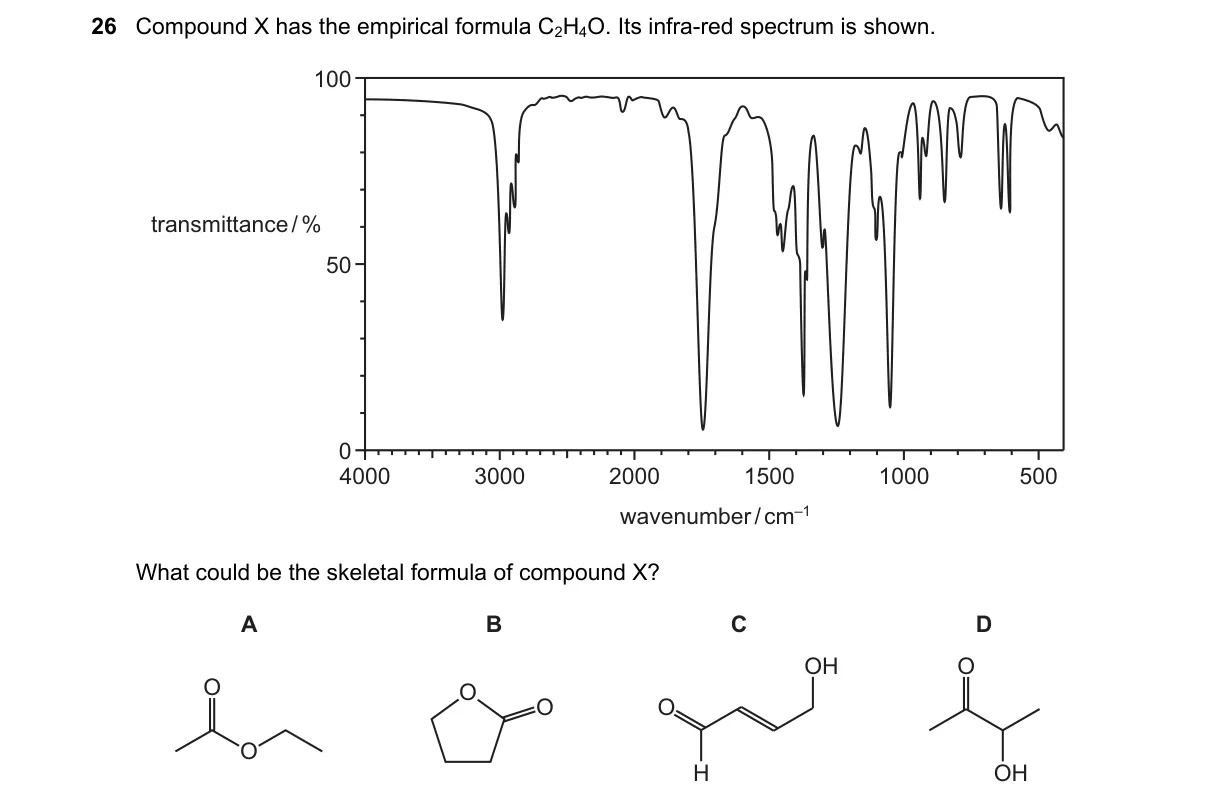
Explanation
IR spectrum identifies carbonyl group in C2H4O isomers
Steps:
- Empirical formula C2H4O matches acetaldehyde, oxirane, or vinyl alcohol.
- IR spectrum shows strong C=O stretch at ~1720 cm⁻¹, indicating aldehyde.
- No O-H stretch (~3400 cm⁻¹) rules out alcohols or enols.
- Absence of C=C stretch (~1650 cm⁻¹) confirms saturated carbonyl.
Why A is correct:
- Acetaldehyde's skeletal formula CH₃-CHO matches C=O IR peak per Hooke's law for carbonyl vibration.
Why the others are wrong:
- B (oxirane): Lacks carbonyl; shows C-O stretch ~1000 cm⁻¹ instead.
- C (ethanol, but wrong formula): Shows broad O-H stretch absent here.
- D (ethenyl alcohol): Unstable enol with O-H and C=C peaks not matching.
Final answer: A
Topic: Analytical techniques
Practice more A Levels Chemistry (9701) questions on mMCQ.me