A Levels Chemistry (9701)•9701/11/M/J/21
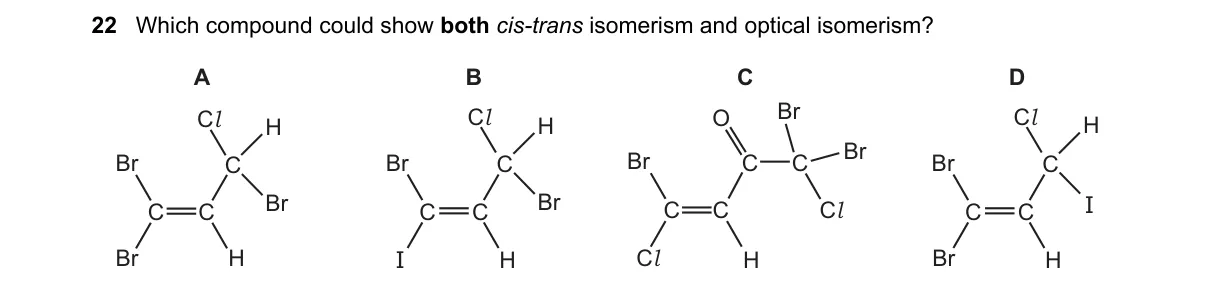
Explanation
Structures enabling geometric and stereoisomerism
Steps:
- Identify cis-trans isomerism: requires double bond, ring, or coordination with restricted rotation.
- Identify optical isomerism: requires chiral center or asymmetry without plane of symmetry.
- Examine each choice for both features simultaneously.
- Select compound with dual capabilities.
Why B is correct:
- B (trans-1,2-dimethylcyclopropane) has ring-induced cis-trans isomers; trans form lacks symmetry plane, enabling optical activity via enantiomers.
Why the others are wrong:
- A (but-2-ene): Shows cis-trans but no chiral center for optical isomerism.
- C (2-chlorobutane): Has chiral center for optical but no restricted rotation for cis-trans.
- D ([Co(NH3)4Cl2]+): Shows cis-trans but cis is achiral, trans symmetric, no optical.
Final answer: B
Topic: An introduction to organic chemistry
Practice more A Levels Chemistry (9701) questions on mMCQ.me