A Levels Chemistry (9701)•9701/13/M/J/20
Explanation
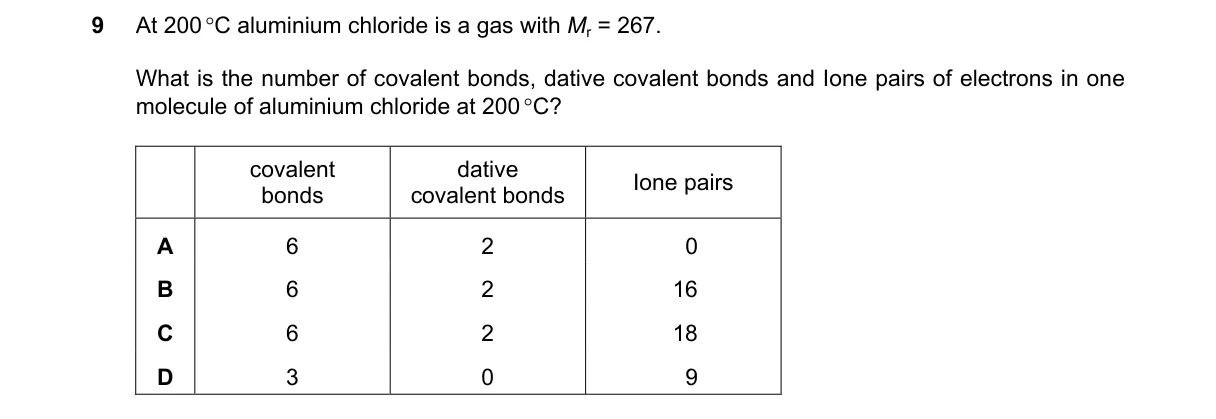
Al2Cl6 Dimer Structure and Bonding Steps:
- Molecular mass M=267 confirms Al2Cl6 (2×27 + 6×35.5 = 267), not monomeric AlCl3 (133.5).
- In Al2Cl6, each Al connects to four Cl: four terminal Al–Cl bonds are covalent; two bridging Cl form two additional covalent bonds and two dative bonds (Cl → Al).
- Total: 6 covalent bonds (4 terminal + 2 bridge) and 2 dative bonds.
- Each of 6 Cl atoms contributes 3 lone pairs (7 valence e – 1 for bonding = 6 e or 3 pairs), totaling 18 lone pairs.
Why B is correct:
- Matches Al2Cl6 bonding model: 6 shared-electron covalent bonds, 2 coordinate dative bonds, and 18 lone pairs on Cl atoms per Lewis structure.
Why the others are wrong:
- A: Incorrectly states 0 lone pairs; Cl atoms have lone pairs to achieve octet.
- C: Duplicate of B; no distinction.
- D: Describes monomeric AlCl3 (3 covalent bonds, 9 lone pairs), but M=267 indicates dimer.
Final answer: B
Topic: Chemical bonding
Practice more A Levels Chemistry (9701) questions on mMCQ.me