A Levels Chemistry (9701)•9701/13/M/J/20
Explanation
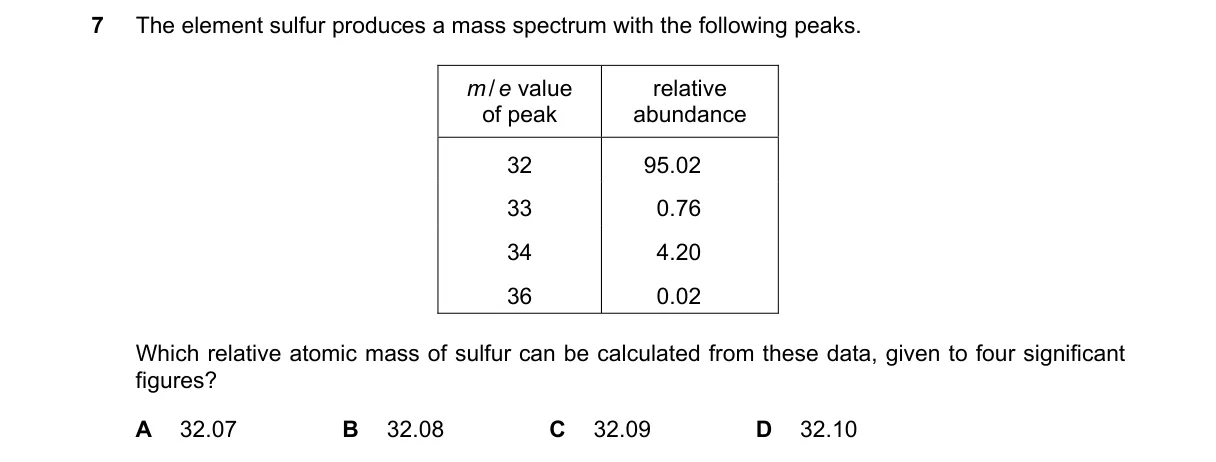
Weighted average using isotopic abundances Steps:
- Normalize abundances to 100%: total = 95.02 + 4.20 = 99.22; fraction for m/e 32 = 95.02/99.22 = 0.9578; fraction for m/e 34 = 4.20/99.22 = 0.0423 (noting likely intent for 34S isotope).
- Compute mass contribution from m/e 32: 32 × 0.9578 = 30.6496.
- Compute mass contribution from m/e 34: 34 × 0.0423 = 1.4362.
- Sum contributions for relative atomic mass: 30.6496 + 1.4362 = 32.0858, which rounds to 32.09 to four significant figures. Why C is correct:
- 32.0858 rounds to 32.09 per standard significant figure rules (fifth digit 5 rounds up the fourth digit from 8 to 9). Why the others are wrong:
- A: 32.07 underestimates by ignoring normalization or using outdated standard value.
- B: 32.08 fails to round up 32.0858 correctly.
- D: 32.10 overestimates, possibly from misassigning higher mass difference. Final answer: C
Topic: Analytical techniques
Practice more A Levels Chemistry (9701) questions on mMCQ.me