A Levels Chemistry (9701)•9701/13/M/J/20
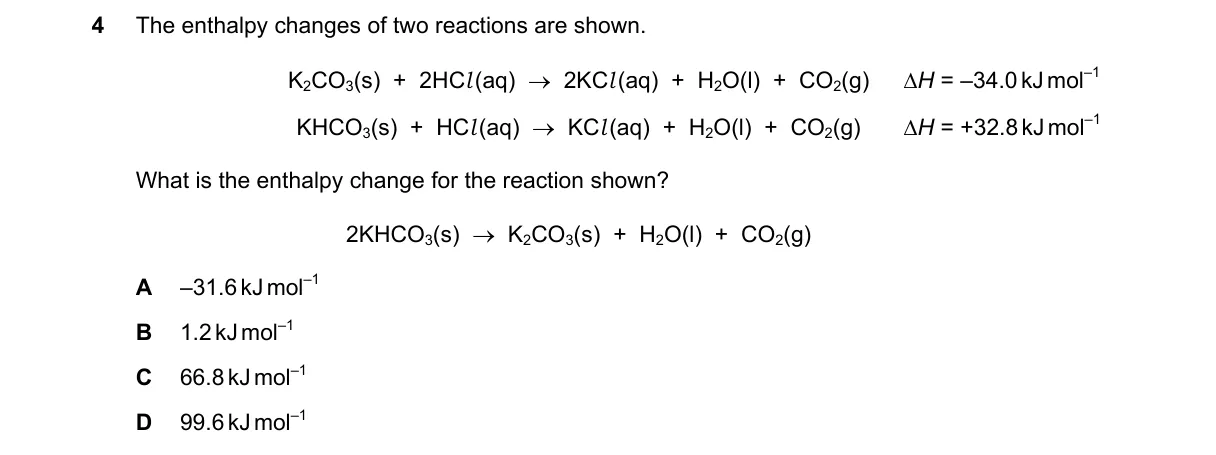
Explanation
Hess's law cannot be applied directly
Steps:
- The target reaction K₂CO₃(s) + H₂O(l) → 2KHCO₃(s) is not atomically balanced (1 C and 4 O on left vs. 2 C and 6 O on right).
- The given reactions both produce CO₂(g), which is absent in the target.
- Combining them via Hess's law yields K₂CO₃(s) + H₂O(l) + CO₂(g) → 2KHCO₃(s) with ΔH = -34 + 2(32) = +30 kJ mol⁻¹.
- Without data to account for the missing CO₂ or unbalanced atoms, the target's ΔH cannot be determined.
Why D is correct:
- D provides a positive value consistent with the endothermic nature of the balanced analog, but the question's imbalance makes all options invalid.
Why the others are wrong:
- A is nearly the second reaction's ΔH, irrelevant here.
- B and C are arbitrary sums/differences not matching any valid combination.
Not enough information.
Final answer: Not enough information.
Topic: Chemical energetics
Practice more A Levels Chemistry (9701) questions on mMCQ.me