A Levels Chemistry (9701)•9701/12/M/J/20
Explanation
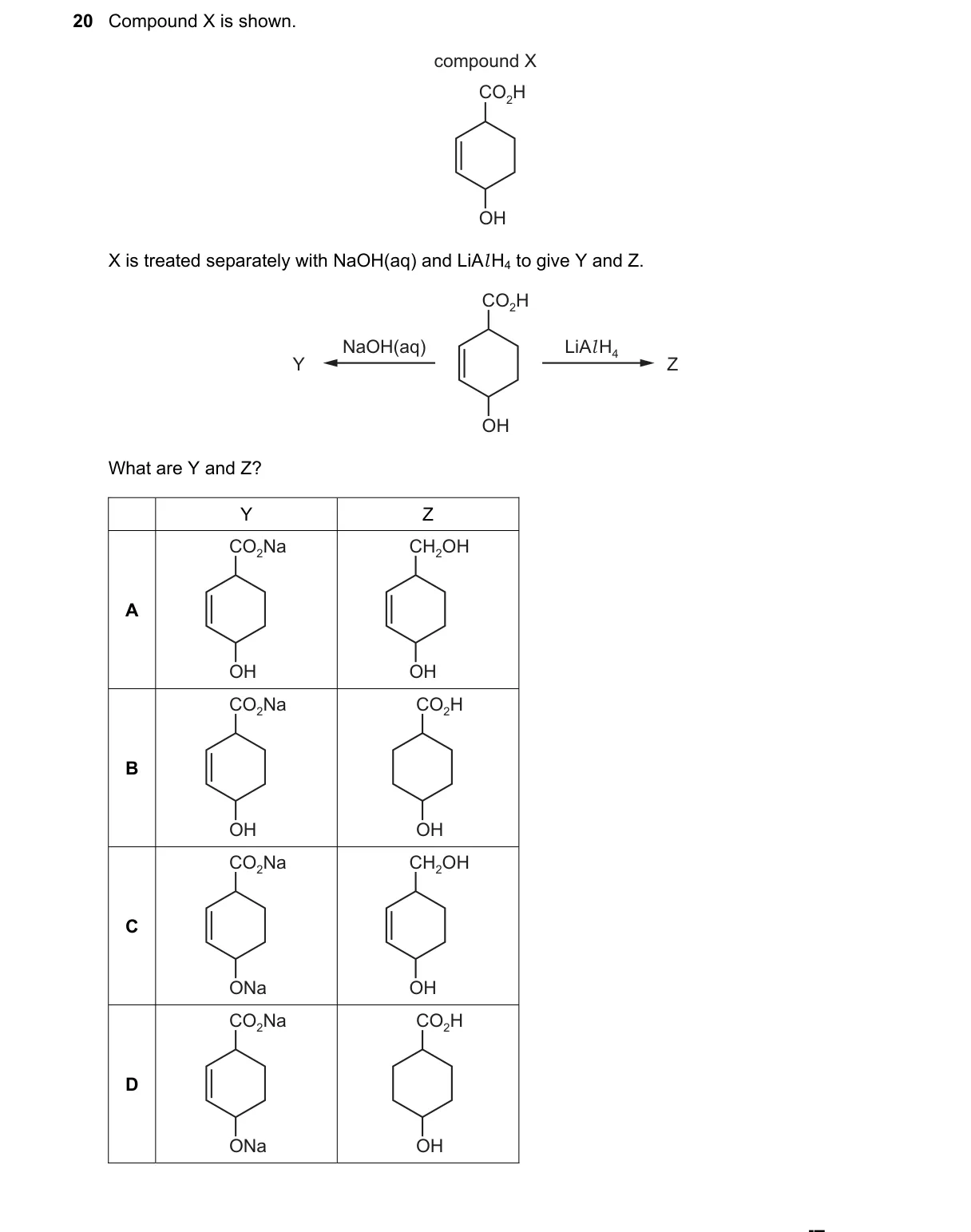
Selective functional group transformations with NaOH and LiAlH4
Steps:
- Compound X contains -COOH and -OH groups.
- NaOH(aq) deprotonates -COOH to -COONa; -OH groups remain unchanged, yielding Y.
- LiAlH4 reduces -COOH to -CH2OH; -OH groups remain unchanged, yielding Z as a polyol.
- Option A depicts Y as the carboxylate salt with intact -OH and Z as the corresponding reduced diol.
Why A is correct:
- NaOH selectively forms the sodium carboxylate from -COOH per acid-base reaction; LiAlH4 reduces -COOH to -CH2OH via hydride addition without affecting alcohols.
Why the others are wrong:
- B: Z shows -COH (aldehyde), but LiAlH4 fully reduces -COOH to -CH2OH, not aldehyde.
- C: Y incorrectly has -ONa on alcohol; NaOH(aq) does not deprotonate alcohols under these conditions.
- D: Combines B and C errors, with -ONa on alcohol and incomplete reduction in Z.
Final answer: A
Topic: Carboxylic acids and derivatives
Practice more A Levels Chemistry (9701) questions on mMCQ.me