Explanation
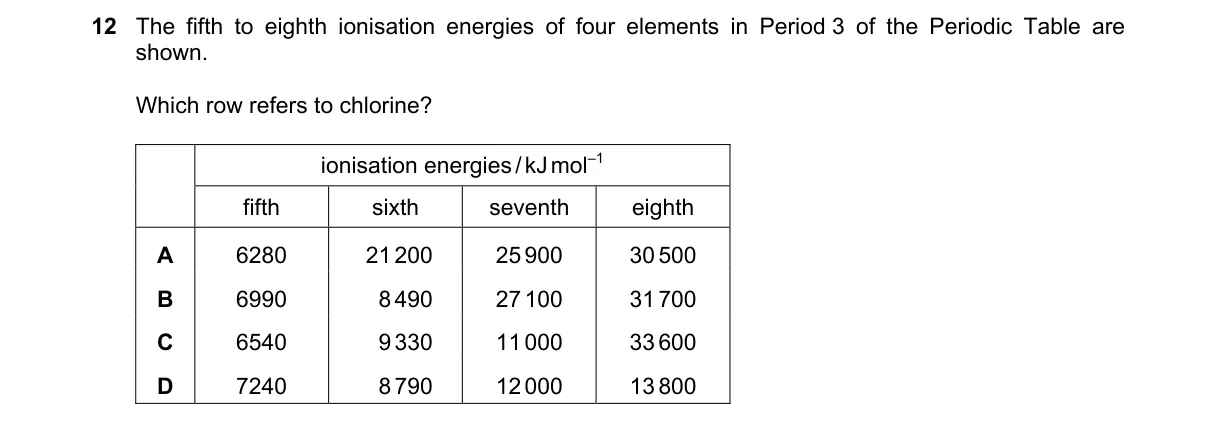
Ionization energy patterns reveal subshell stability in Period 3 elements Steps: - Identify Period 3 elements' valence configurations: chlorine (Cl) has [Ne] 3s² 3p⁵, so fifth IE removes last 3p electron, sixth and seventh remove 3s electrons (all valence, moderate increases), eighth accesses core 2p (sharp rise). - Compare options: look for three successive moderate IEs (fifth–seventh) followed by a large jump to eighth. - Option C shows 8540, 9430, 11000 (small rises), then 31700 (huge jump), matching Cl's pattern. - Eliminate others: A jumps early at sixth; B jumps at seventh; D shows steady increases without core jump. Why C is correct: - Matches halogen trend where valence shell removal (fifth–seventh IEs) yields gradual increases per Aufbau principle, but core penetration (eighth IE) causes exponential rise due to higher effective nuclear charge on inner orbitals. Why the others are wrong: - A: Premature jump at sixth IE fits aluminum (post-p subshell completion). - B: Jump at seventh IE aligns with sulfur (earlier core access after 3s²). - D: No significant jump indicates magnesium (all inner s/p electrons removed gradually). …
Practice more A Levels Chemistry (9701) questions on mMCQ.me