A Levels Chemistry (9701)•9701/11/M/J/20
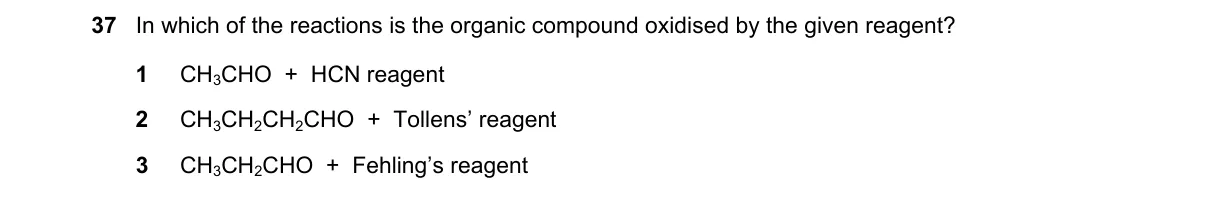
Explanation
Oxidation involves increase in oxygen or loss of electrons in organic compounds
Steps:
- Identify oxidation as a reaction where the organic substrate loses electrons or gains oxygen, often tested by specific reagents like Tollens' or Fehling's for carbonyls.
- Examine each option: Check if the reagent typically oxidizes the given functional group.
- Rule out non-oxidative reactions like additions.
- Confirm the reaction that changes the oxidation state of the carbon atom.
Why B is correct:
- Tollens' reagent oxidizes aldehydes (RCHO) to carboxylic acids (RCOOH), as per the reaction: RCHO + 2[Ag(NH3)2]+ + 3OH- → RCOONH4 + 2Ag + 4NH3 + 2H2O, increasing the aldehyde carbon's oxidation state from -1 to +3.
Why the others are wrong:
- A: HCN adds to the carbonyl of CH3CHO to form a cyanohydrin (nucleophilic addition), no change in oxidation state.
- C: Fehling's reagent does not oxidize alcohols like CH3CH2OH; it requires an aldehyde group for reduction of Cu2+ to Cu2O.
- D: Not enough information (option incomplete).
Final answer: B
Topic: Carbonyl compounds
Practice more A Levels Chemistry (9701) questions on mMCQ.me