A Levels Chemistry (9701)•9701/11/M/J/20
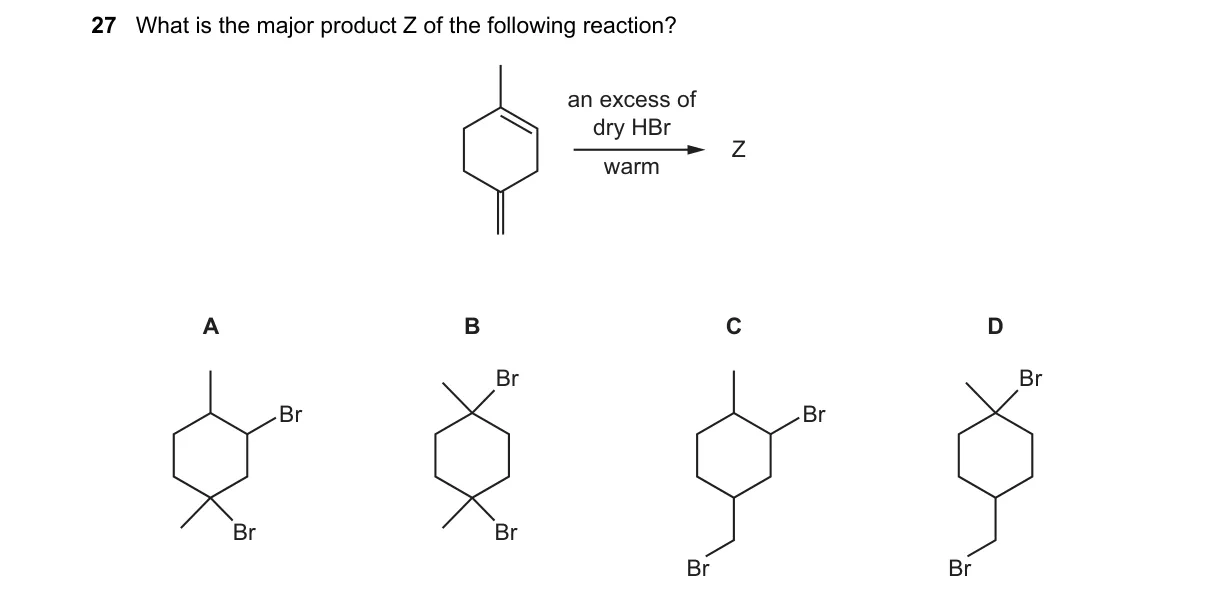
Explanation
Markovnikov double addition to terminal alkyne
Steps:
- Protonate the triple bond of the terminal alkyne to form the more stable vinyl carbocation (Markovnikov orientation).
- Bromide adds to the carbocation, yielding a vinyl bromide intermediate.
- With excess HBr, the alkene in the intermediate undergoes a second protonation to form a secondary carbocation.
- Second bromide addition gives the geminal dibromide as the major product.
Why B is correct:
- B is the geminal dibromide (e.g., CH3-CHBr2 for propyne), following Markovnikov's rule where both Br atoms add to the same carbon.
Why the others are wrong:
- A is the vinyl monobromide, incorrect as excess HBr drives double addition.
- C is the anti-Markovnikov vinyl bromide, violating regioselectivity without peroxides.
- D is the vicinal dibromide, incorrect as additions to alkynes favor geminal over vicinal products.
Final answer: B
Topic: Hydrocarbons
Practice more A Levels Chemistry (9701) questions on mMCQ.me