A Levels Chemistry (9701)•9701/11/M/J/20
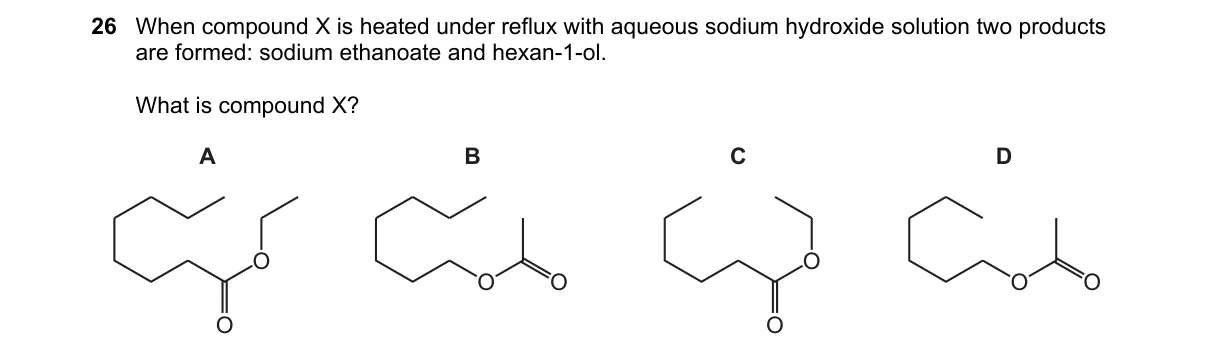
Explanation
Hydrolysis of mixed dialkyl sulfate Steps:
- Identify the reaction conditions: aqueous NaOH under reflux suggests nucleophilic cleavage producing alcohols.
- Observe products: two alcohols (C₂H₅OH and C₆H₁₃OH) indicate X contains both ethyl and hexyl groups linked to a group displaceable by OH⁻.
- Recall dialkyl sulfates hydrolyze with NaOH: (RO)(R'O)SO₂ + 2NaOH → ROH + R'OH + Na₂SO₄.
- Conclude X is ethyl hexyl sulfate, matching the alkyl groups to the products.
Why D is correct:
- D is ethyl hexyl sulfate, (C₂H₅O)(C₆H₁₃O)SO₂, which hydrolyzes via double nucleophilic substitution to yield exactly ethanol and hexan-1-ol.
Why the others are wrong:
- A (ethyl hexanoate): Yields ethanol and sodium hexanoate, not hexan-1-ol.
- B (hexyl ethanoate): Yields hexan-1-ol and sodium ethanoate, not ethanol.
- C (ethyl hexyl ether): Unreactive to aqueous NaOH; no cleavage to alcohols.
Final answer: D
Topic: Carboxylic acids and derivatives
Practice more A Levels Chemistry (9701) questions on mMCQ.me