A Levels Chemistry (9701)•9701/13/M/J/19
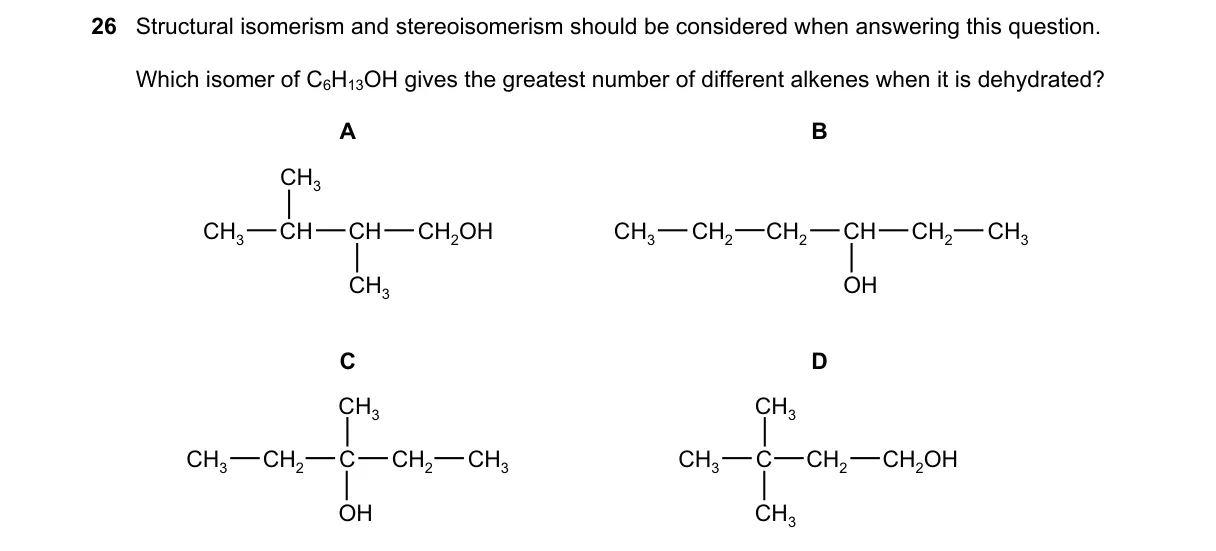
Explanation
Carbocation stability in secondary alcohols maximizes alkene diversity in dehydration.
Steps:
- Classify isomers: A (2-methylpropan-1-ol, primary), B (butan-2-ol, secondary), C (butan-1-ol, primary), D (2-methylpropan-2-ol, tertiary).
- Primary alcohols (A, C) dehydrate mainly via E2, producing one alkene each without stereoisomers.
- Secondary (B) and tertiary (D) dehydrate via E1, forming carbocations; B's is unsymmetric, D's symmetric.
- For B, CH3CH2CH+CH3 loses H+ from methyl (1-butene) or methylene (cis-/trans-2-butene), yielding three alkenes.
Why B is correct:
- Unsymmetric secondary carbocation enables elimination to one constitutional isomer without stereo (1-butene) and one with E/Z stereoisomers (2-butene), per E1 mechanism rules.
Why the others are wrong:
- A: E2 or rearranged E1 yields only 2-methylpropene (no stereo).
- C: E2 yields only 1-butene (no stereo; minor E1 ignored for primary).
- D: Symmetric tertiary carbocation yields only 2-methylpropene (equivalent betas, no stereo).
Final answer: B
Topic: Hydroxy compounds
Practice more A Levels Chemistry (9701) questions on mMCQ.me