A Levels Chemistry (9701)•9701/13/M/J/19
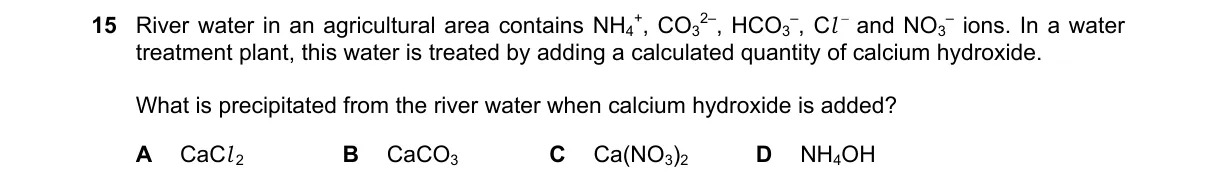
Explanation
Precipitation of Insoluble Calcium Carbonate Steps:
- River water contains NH₄⁺, HCO₃⁻, Cl⁻, and NO₃⁻ ions; calcium hydroxide (Ca(OH)₂) dissociates to Ca²⁺ and OH⁻.
- OH⁻ reacts with HCO₃⁻ to form CO₃²⁻ and H₂O: HCO₃⁻ + OH⁻ → CO₃²⁻ + H₂O.
- Ca²⁺ combines with CO₃²⁻ to form CaCO₃, which is insoluble and precipitates.
- Other ions (NH₄⁺, Cl⁻, NO₃⁻) form soluble compounds with Ca²⁺ or OH⁻, so no other precipitates occur.
Why B is correct:
- CaCO₃ has low solubility (Ksp = 3.3 × 10⁻⁹), forming a precipitate per solubility product rules.
Why the others are wrong:
- A: CaCl₂ is highly soluble in water.
- C: Ca(NO₃)₂ is soluble, used in fertilizers.
- D: NH₄OH is a soluble weak base, not a solid precipitate.
Final answer: B
Topic: Group 2
Practice more A Levels Chemistry (9701) questions on mMCQ.me