A Levels Chemistry (9701)•9701/12/M/J/19
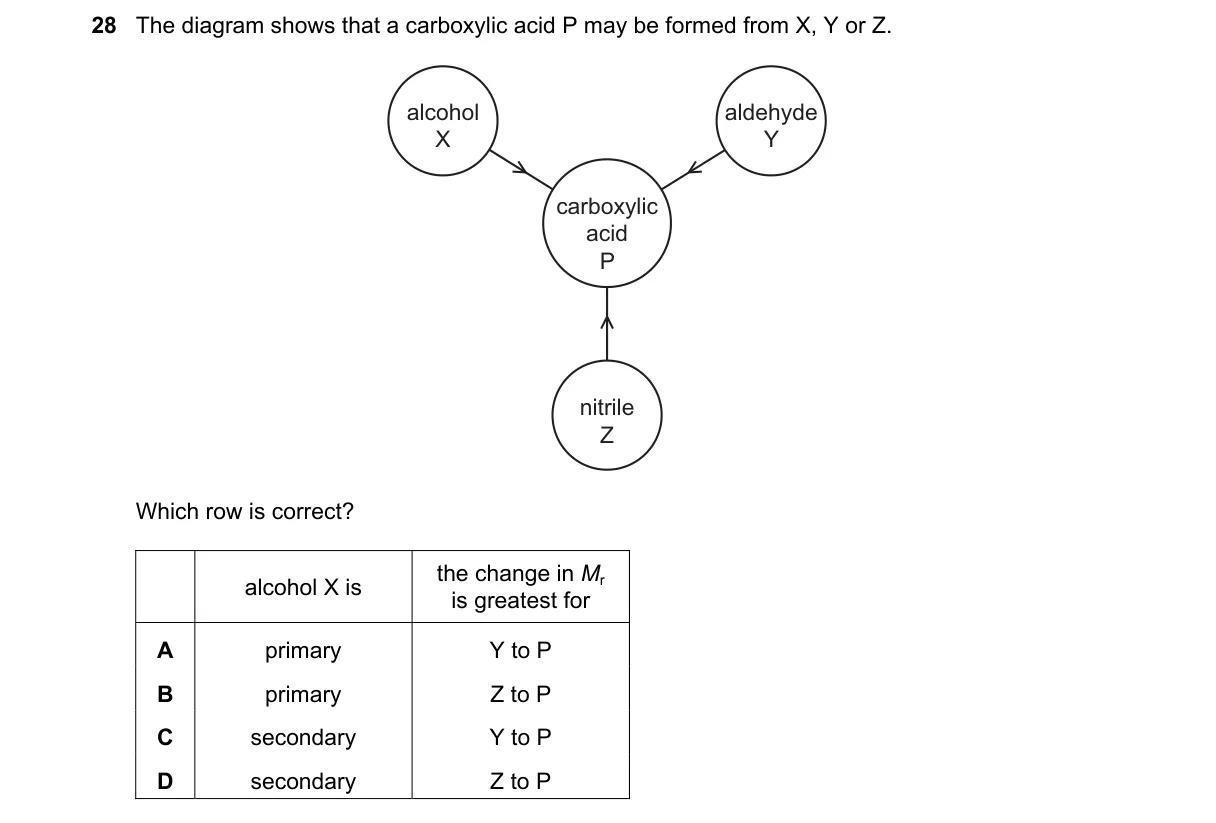
Explanation
Primary alcohol oxidation to carboxylic acid with molar mass comparison
Steps:
- Carboxylic acid P forms via oxidation, which requires a primary alcohol starting material, as secondary alcohols yield ketones.
- X is labeled as an alcohol, so it must be primary to produce P.
- Y and Z are alternative precursors (e.g., aldehyde or alkene); compare molar masses (Mr) from diagram.
- Greatest ΔMr occurs for Z to P, as Z has the lowest Mr, leading to largest increase upon forming P.
Why B is correct:
- Primary alcohols oxidize to carboxylic acids (RCH₂OH → RCOOH, ΔMr = +14 via O gain, 2H loss), enabling X → P; Z → P shows max ΔMr per diagram.
Why the others are wrong:
- A: ΔMr greatest for Y to P, but diagram shows Z to P larger.
- C: X secondary cannot oxidize to carboxylic acid, violating formation rule.
- D: Combines secondary X error with wrong ΔMr path (Y instead of Z).
Final answer: B
Topic: Hydroxy compounds
Practice more A Levels Chemistry (9701) questions on mMCQ.me