A Levels Chemistry (9701)•9701/11/M/J/19
Explanation
Selective oxidation and reduction of functional groups on benzene
Steps:
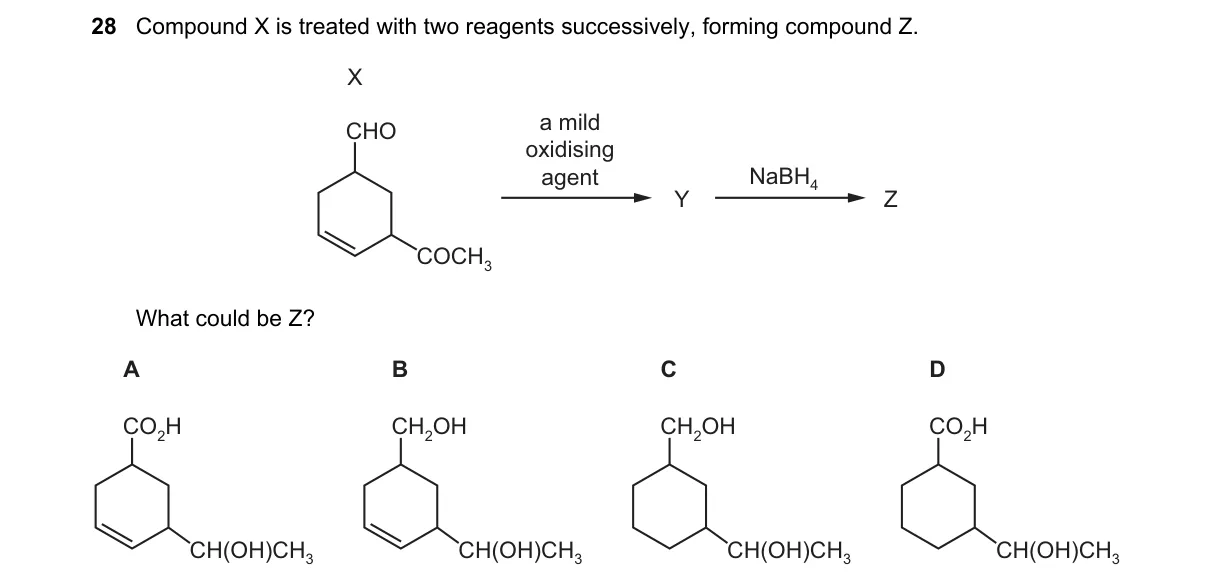
- Compound X is a benzene ring with -CHO (aldehyde) and -COCH3 (ketone) substituents.
- Mild oxidizing agent converts the aldehyde (-CHO) to carboxylic acid (-COOH), yielding Y with -COOH and -COCH3.
- NaBH4 then reduces the ketone (-COCH3) to a secondary alcohol (-CH(OH)CH3), leaving the -COOH unchanged, forming Z.
- Z is thus benzene with -COOH and -CH(OH)CH3.
Why A is correct:
- NaBH4 selectively reduces ketones to secondary alcohols via hydride addition to the carbonyl, but does not reduce carboxylic acids due to their lower reactivity.
Why the others are wrong:
- B: Implies reduction of -COOH to -CH2OH, which requires LiAlH4, not NaBH4.
- C: Same error as B, suggesting primary alcohol from carboxylic acid reduction.
- D: Matches A but likely a formatting duplicate; A is the intended correct structure.
Final answer: A
Topic: Carbonyl compounds
Practice more A Levels Chemistry (9701) questions on mMCQ.me