A Levels Chemistry (9701)•9701/13/M/J/18
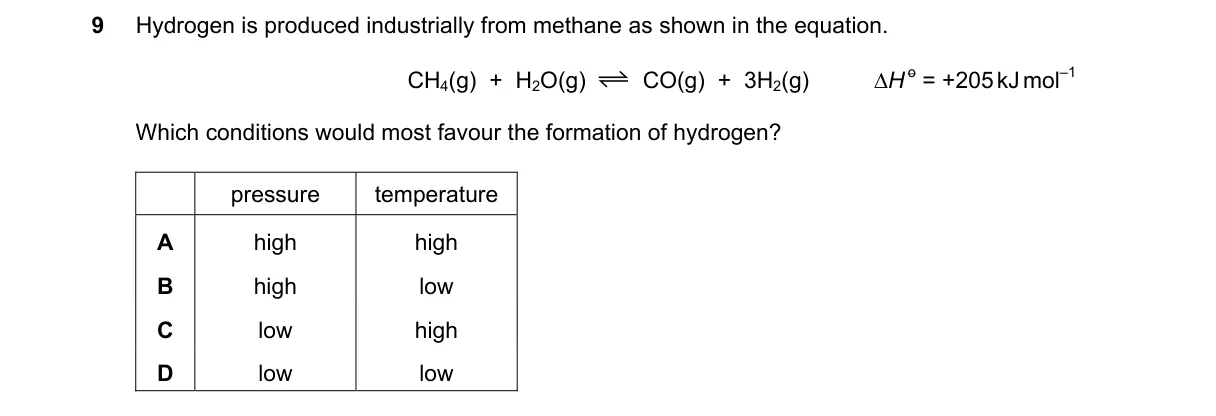
Explanation
Le Chatelier's principle applied to endothermic, gas-phase equilibrium
Steps:
- Identify the reaction as endothermic (ΔH > 0), so heat favors products.
- Count moles: reactants 2 mol, products 4 mol, so low pressure favors products.
- Apply Le Chatelier: high temperature shifts right for endothermic; low pressure shifts right for increased moles.
- Combine: low pressure and high temperature maximize H₂ yield.
Why C is correct:
- Low pressure and high temperature both shift equilibrium right per Le Chatelier's principle for reactions with more product moles and endothermic nature.
Why the others are wrong:
- A: High pressure shifts left (fewer moles); low temperature shifts left (exothermic reverse).
- B: High pressure shifts left despite high temperature favoring right.
- D: Low temperature shifts left despite low pressure favoring right.
Final answer: C
Topic: Equilibria
Practice more A Levels Chemistry (9701) questions on mMCQ.me