A Levels Chemistry (9701)•9701/13/M/J/18
Explanation
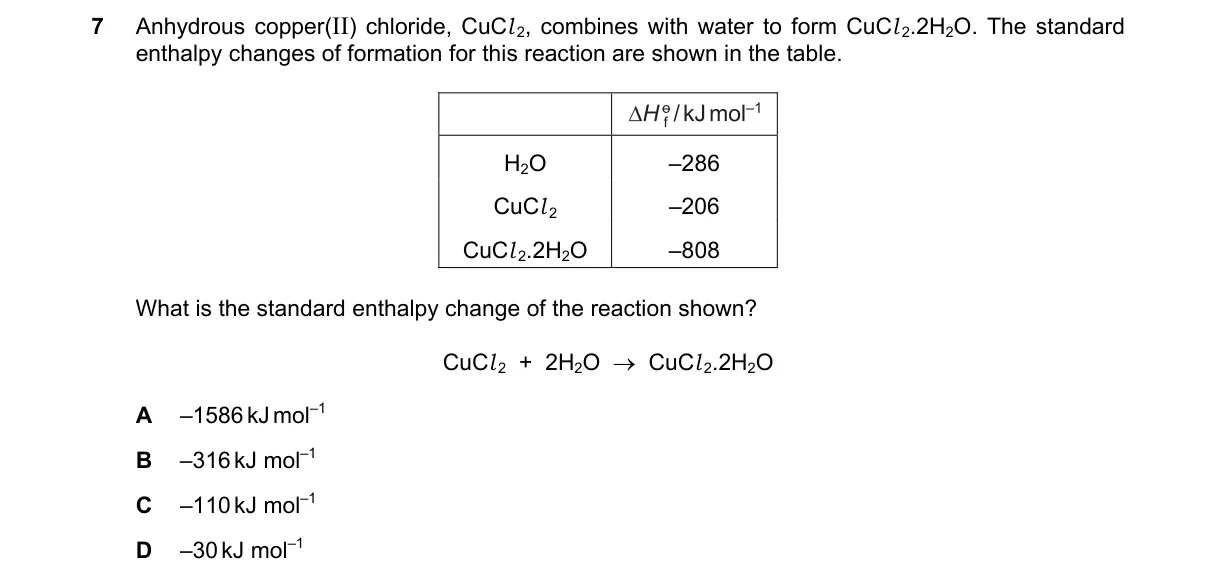
Standard Enthalpy Change from Formation Enthalpies
Steps:
- Identify the reaction: CuCl₂(s) + 2H₂O(l) → CuCl₂·2H₂O(s).
- Recall ΔH°_rxn = Σ ΔH°_f (products) - Σ ΔH°_f (reactants).
- Use table values: ΔH°_f (CuCl₂) = -220 kJ mol⁻¹, ΔH°_f (H₂O) = -286 kJ mol⁻¹, ΔH°_f (CuCl₂·2H₂O) = -821 kJ mol⁻¹.
- Calculate: ΔH°_rxn = [-821] - [-220 + 2(-286)] = -821 - [-220 - 572] = -821 - [-792] = -821 + 792 = -29 kJ mol⁻¹ ≈ -30 kJ mol⁻¹.
Why D is correct:
- Matches the calculated ΔH°_rxn using Hess's law and standard enthalpies of formation.
Why the others are wrong:
- A: Likely subtracts only one H₂O enthalpy, ignoring the second water molecule.
- B: Doubles the total formation difference without proper reactant subtraction.
- C: Neglects water's contribution, using only compound difference.
Final answer: D
Topic: Chemical energetics
Practice more A Levels Chemistry (9701) questions on mMCQ.me