A Levels Chemistry (9701)•9701/13/M/J/18
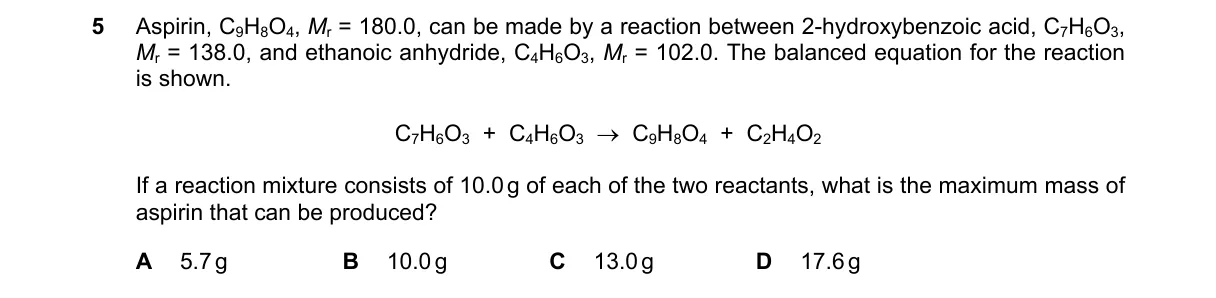
Explanation
Limiting reactant calculation for aspirin yield
Steps:
- Moles of 2-hydroxybenzoic acid: 10.0 g / 138 g/mol = 0.0725 mol.
- Moles of ethanoic anhydride: 10.0 g / 102 g/mol = 0.0980 mol.
- Acid is limiting (1:1 ratio, fewer moles than anhydride).
- Maximum aspirin: 0.0725 mol × 180 g/mol = 13.0 g.
Why C is correct:
- 13.0 g matches mass from limiting reactant's moles times aspirin's molar mass (stoichiometric yield formula).
Why the others are wrong:
- A: Underestimates by ignoring full conversion of limiting reactant.
- B: Assumes 1:1 mass ratio, ignores molar masses and stoichiometry.
- D: Overestimates using anhydride moles, which is in excess.
Final answer: C
Topic: Carboxylic acids and derivatives
Practice more A Levels Chemistry (9701) questions on mMCQ.me